Unispray

Product Description
Nemera

-
FR
-
2015On CPHI since
-
1Certificates
-
1000 - 4999Employees
Company types
Categories
Nemera

-
FR
-
2015On CPHI since
-
1Certificates
-
1000 - 4999Employees
Company types
More Products from Nemera (25)
-
Product Safe’n’Spray(TM): Managing drug overdosing easily with a smart electronic concept device
Safe'n'Spray: the smart electronic concept device with locking features Some drugs such as strong painkillers called opioids (e.g. Fentanyl) can cause respiratory depression in case of overdose, and can be fatal. That’s why patients must rely on a device with locking and child-resistant features for multid... -
Product Sof’Bag®+ protects the formulation and delivers it on a reliable manner
Sof’Bag®+, the airless solution for your sensitive formulations Sof’Bag®+ is an airless pouch-in-bottle technology allowing the formulation to be dispensed out of a collapsing pouch encased inside a rigid bottle upon pump actuation. Each actuation delivers a precise metered dose of formulation, which is di... -
Product Symbioze(TM): smart and sustainable on-body injector platform to improve patients’ injection experience
Symbioze: fostering patient adherence with high-volume, multiple-use wearable. Symbioze is specifically tailored to administer complex drugs, such as biologic therapies, thanks to a highly engineered, reliable drug delivery system. It is suitable for large volumes such as 20ml and beyond, while preserving... -
Product UniSpray: single-metered dose for systemic-acting drug administration
UniSpray: ideal systemic delivery for emergency lifesaving and crisis treatments UniSpray is a unidose system concept under development, designed to be robust and compatible with existing marketed primary container. Through usability analysis and Freedom to Operate assessment, we assure reliability and reg... -
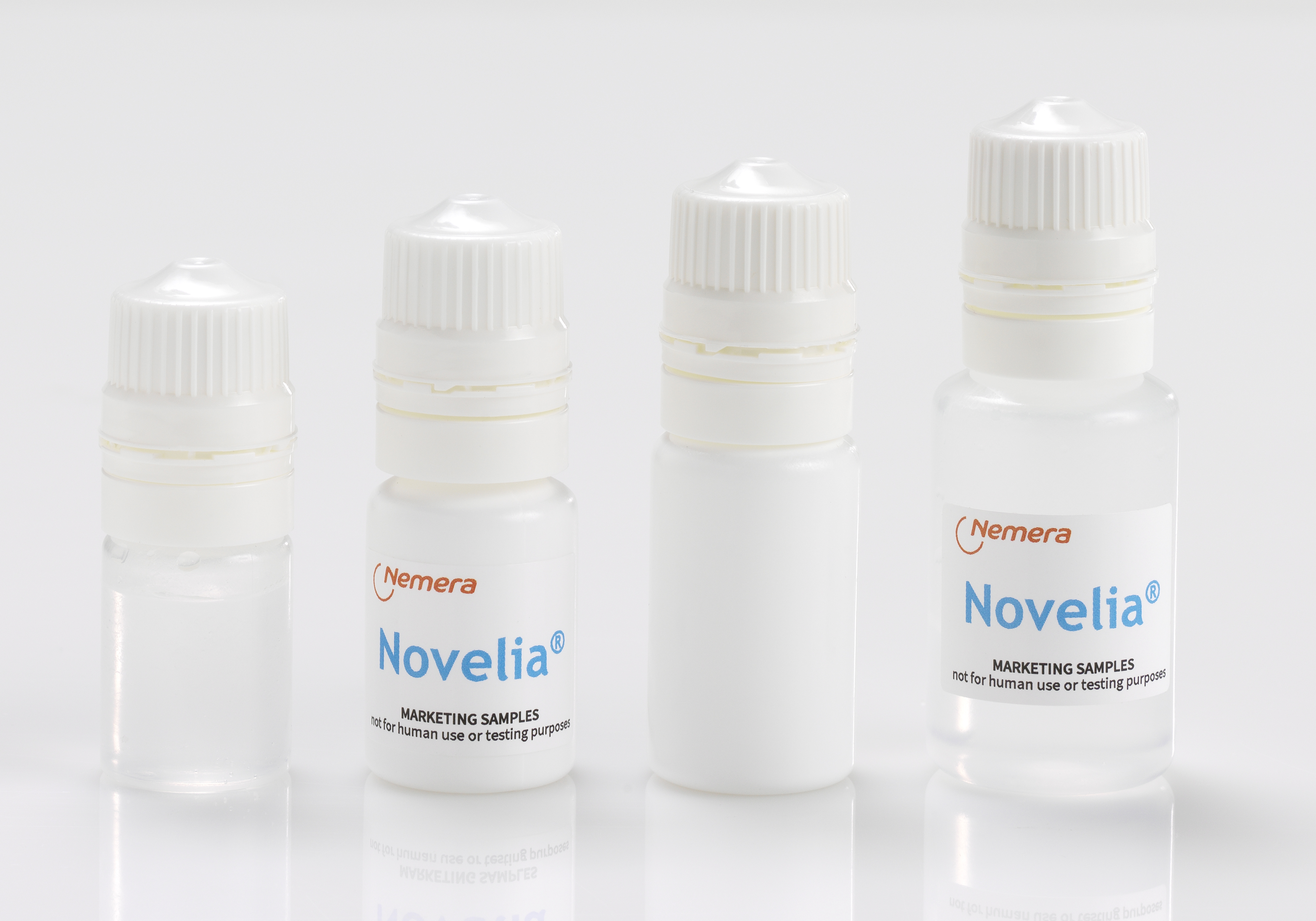
Product Novelia®: Nemera’s preservative-free multidose eyedropper
Novelia®: Nemera’s preservative-free multidose eyedropper, is used across 4 continents
Preservatives have been shown to be harmful to the eye and can lead patients to discontinue or skip treatment to avoid irritation and discomfort.
The new preservative-free multidose ophthalmic packaging Nove... -
Product PUMPS SP27 / SP270+: quality and performance for ENT sprays
Nemera offers an extensive range of metered pumps and valves, compatible with a wide choice of actuators in different shapes and fitments for nasal, buccal and auricular delivery.
Here our different pumps:
SP27 - SP 37: Quality systems for unregulated markets
SP270+ - SP370+: Qual... -
Product Safe'n'Spray
Safe’n’Spray, the new smart nasal spray with locking features, dramatically improves the existing way of administrating specific pain treatments, avoiding the risk of overdosing.
-
Product DPIs / PMDIs : expert in development & manufacturing of respiratory drug delivery devices
Nemera has a recognized expertise in the development and manufacturing of respiratory drug delivery devices, such as Dry Powder Inhalers (DPIs), pressurized Metered Dose Inhalers (pMDIs) and dose counters.
More than ten million asthmatics rely everyday on devices manufactured by Nemera.
-
Product DPI / PMDI
From early-stage concept development to manufacturing we are truly a holistic partner in inhalation.
With a long-standing know-how in inhalation delivery, we are today the leader in the Dry Powder Inhalers (DPIs) manufacturing business.
Our expertise covers Dry Powder Inhalers (reservoir, b... -
Product Safe'n'Sound®
Safe’n’Sound® (1ml & 2.25ml) is a highly customizable platform of add-on passive safety devices for prefilled syringes. It helps in preventing needlestick injuries, providing user-friendly protection for healthcare professionals and patients or caregivers.
Ergonomically designed, Safe’n’Sound® is ... -
Product Symbioze
Symbioze is specifically tailored to administer complex drugs, such as biologic therapies, thanks to a highly engineered, reliable drug delivery system. It is suitable for various drug platforms and compatible with market-proven cartridges, as well as standard manufacturing processes. The cartridge is pr... -
Product Pen Vario
Our portfolio of disposable pens includes variable and fixed dose devices for automatic and manual drug administration, combining modern design with high intuitiveness of use for multidose therapies.
PENVARIO – Dedicated to manufacturing of disposable, variable-dose pen injectors
Nemera resources (22)
-
News Nemera: Stronger than ever
More modern, our new brand identity truly embodies who we are, how we partner with our customers and ultimately the value we bring to millions of patients across the world through our drug delivery device solutions. -
Video Innovation in Connectivity Roundtable pt.1
The Innovation in Connectivity Roundtable originally aired as part of Pharmapack Europe 2021 Part 1 of this session includes three presentations: Patient Journey Driven Considerations for Integrating Digital Health for Combination Products The importance of utilizing applied ethnography and other primary research methods to understand the patient journey as the foundation for integrating digital health into a device/combination product development initiative. Considering patient needs in the context of a larger ecosystem including providers, payers, and regulators to develop a robust plan with respect to connectivity and digital health. Examples of how this understanding is applied across the combination product development journey through organic development, utilization of platform devices, or as a lifecycle management enchantments while optimizing the user experience resulting in safe, effective, and differentiated combination products. Mark Tunkel, Global Category Director for Insight Innovation, Nemera Electronic Product Information: A Case Study on Medicine Labelling for the Visually Impaired The Electronic Product Information Initiative for Medicines (ePI) aim to ease the accessibility of product information to everyone, including visually impaired patients. The GS1 Digital Link aim to provide a single barcode for identification, brand protection, and access to product information for patients. Our lecture will illustrate how the existing Datamatrix on packaging could be leveraged, today, by everyone, including visually impaired users, to access a tailored and relevant information. Laurent Tonnelier, CEO, mobiLead - xTag Lise Wagner, Accessibility Specialist and User Expert, OKEENEA Traceability of Medicines: from Finished Product to Unit Dose Delivery Challenges for B2B Supply Chain stakeholders Regulations: a global overview What about B2C Nathalie Wardé, CEO & Founder, D4P pharma Join the above speakers and moderator Lionel Jeannin, Group Head Oral Packaging & Delivery Systems, Novartis Pharma AG for the live panel discussion that followed in the Innovation in Connectivity Roundtable pt. 2 -
News Pharmapack Europe 2021 announces Awards winners!
Awards showcased a wide range of industry-changing innovations in packaging and drug delivery, with sustainability emerging as a key metric across all categories -
Video Innovation in Connectivity Roundtable pt. 2
The second part of the Innovation in Connectivity Roundtable brings together presenters from Nemera, D4P Pharma and xTag for a group discussion and live Q&A (45-minutes). Moderated by Lionel Jeannin, Group Head Oral Packaging & Delivery Systems, Novartis Pharma AG Participants: Mark Tunkel, Global Category Director for Insight Innovation, Nemera Laurent Tonnelier, CEO, mobiLead – xTag Lise Wagner, Accessibility Specialist and User Expert, OKEENEA Nathalie Wardé, CEO & Founder, D4P pharma This webinar originally aired as part of Pharmapack 2021 -
News Pharmapack 2021: Increasing connectivity is the key to patient centricity and simplified access to product information
During the live Innovation in Connectivity Roundtable at the Pharmapack 2021 online conference, experts discussed how the latest innovations in digital technology are shaping industry ambitions to put the patient journey at the heart of its drug device and delivery solutions by simplifying product information via increased connectivity. -
Video Welcome to the Digital Health & Connectivity & Patient-Centricity Track
This webinar will address: Key market trends in digital health and how they have accelerated in the past years Nemera’s vision of digital health and the implications on the whole drug/device development ecosystem – how connectivity is creating incredible opportunities for the industry but also raises some challenges: link to conferences in the track How does Nemera play in this landscape: presentation of the company, our capabilities and some examples of our connected devices. Webinar originally aired as part of Pharmapack Europe 2021 -
News Considerations for device selection in parenteral applications.
While patient centricity is the foundation for developing a robust device selection plan and completion of a successful combination product development initiative, developers need to remember there is a larger ecosystem that must be considered to ensure success.
-
Video Nemera: We Put Patients First
As a world-leading drug delivery device solutions provider, our purpose of putting patients first enables us to design and manufacture devices that maximize treatment efficacy. We are a holistic partner and help our customers succeed in the sprint to market of their combination products. From early device strategy to state-of-the-art manufacturing, we’re committed to the highest quality standards. Agile and open-minded, we work with our customers as colleagues. Together, we go the extra mile to fulfill our mission.
For more information, visit www.nemera.net -
News Nemera is excited to announce that its multidose eye dropper Novelia® has been approved in Brazil!
Nemera is excited to announce that its multidose eye dropper Novelia® has been approved by ANVISA for the preservative-free formulations CLILON [1] (Ketorolac 0,4% and 0,5%), a nonsteroidal anti-inflammatory drug (NSAID) that is used to treat moderately severe pain and inflammation, usually after surgery. -
Video NEMERA CEO SHARES 2021 AMBITION
Our CEO, Marc Hämel shares the lessons we learned from last year, extends his wishes, gratitude to the Nemera family and walks us through the top company goals that will shape and drive our story in 2021! -
News Nemera – an integrated CDMO and Own-IP player
In this report, Alira Health in partnership with MassMEDIC analyzes the rapidly evolving DDD landscape and how industry leaders like Nemera strive to enhance the end-to-end services offering by integrating the pharmaceutical value chain. -
Video Nemera at CPHI Worldwide 2019 - Bernhard Rohn and Mark Tunkel
Our VP strategy and marketing and our director of business development, Insight Innovation Center are presenting Nemera and its portfolio. They are giving more details about Insight Innovation Center. -
News The pivotal role played by device developers to improve patient eye care
Our ophthalmic experts discuss the challenges glaucoma and dry eye patients face with eye drop administration and present the Novelia® system featuring patented PureFlow® technology, which controls the medication flow. -
Video Nemera at CPHI Worldwide 2018
Nemera, Juan Sarmien, Sales Director for North America at Nemera, speaks with CPHI TV at CPHI Worldwide in Madrid. -
News Nemera breaks ground in Brazil through Milfra acquisition
Nemera announced that they have entered into an agreement to acquire Milfra. Milfra, based in Jaguariuna, state of São Paulo, Brazil, specializes in the development and production of both vaginal and rectal applicators, as well as oral dosers for the pharmaceutical industry. A family-owned company f... -
News Nemera expands its insulin pen’s offer in Brazil
This partnership expands offer of diabetes medicines in the country; Nemera’s Advapen launch happens under the trade name Lifepen® for the administration of Glargilin insulin for the treatment of diabetes A pioneer in biotechnological medicines in Brazil, BIOMM is launching Nemera’s Advapen under the trade name Lifepen® for the administration of Glargilin insulin for the treatment of diabetes.
-
Video Nemera: Sronger Than Ever
Our proven experience in the complementary activities surrounding drug delivery device and combination product development, alongside our strong know-how in our industry ecosystem, puts us in an optimal place to support our customers as partners through every step in their journey. By keeping the patient top of mind, we create success on all fronts. We aim to improve patients’ outcomes and build customer success. In order to do that, we have three key pillars that form the base of our organizational identity: • Patients first. Always. • Partnering in drug delivery devices. Holistically. • Going the extra mile. Together. Our new logo and a new brand style convey this message to customers, applicants, shareholders, and other stakeholders, with a fresh, friendly, and moving-forward look and feel. -
News Nemera to acquire Copernicus to boost parenteral product portfolio and small series capabilities
Nemera today announced that they have entered into an agreement to acquire Copernicus. Copernicus, based in Szczecin Poland, specializes in the development and manufacturing of injection devices. Their range of reusable and disposable pen injectors are tailored for the treatment of several chronic pathologies.
-
News Nemera and Noble collaboration for training on Safe’n’Sound® 2.25 ml
As one of the world’s leading providers of design, development and manufacturing of drug delivery devices, we announced the conclusion of a collaboration agreement with Noble, global provider of medical device training solutions and patient onboarding strategies for the world’s top pharmaceutical and biotechnology brands. On this occasion, the latest issue of ‘The Pharmaceutical post’ publish an article about the collaboration between Nemera and Noble. -
News NASAL SPRAY BIOEQUIVALENCE
In the pharmaceutical industry, we can see the development of generics is significantly growing. This trend is mainly driven by countries to give patients easier access to drugs and to define regulations accordingly. -
News RETRONOSE: IMPROVING NASAL DELIVERY THROUGH NEW AND IMPROVED DEVICE
RetroNose is a completely new drug delivery concept to dispense drug formulation to the nasal cavity. The principle of this concept is to deliver a spray through the oral cavity to deposit the drug in the nasal cavity from rear to front. To avoid aerosol penetration in the lung and to ensure deposition efficacy, the aerosol drug is automatically delivered during the nasal expiratory phase.
Frequently Viewed Together
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance



















-file122583.png)

-file122681.png)

-file122584.png)