Transfer Methods Service

Product Description
Avara Pharmaceutical Services
-
US
-
2017On CPHI since
-
2Certificates
-
500 - 999Employees
Company types
Primary activities
Categories
Specifications
Avara Pharmaceutical Services
-
US
-
2017On CPHI since
-
2Certificates
-
500 - 999Employees
Company types
Primary activities
More Products from Avara Pharmaceutical Services (26)
-
Product Product Release Testing Service
Avara Pharmaceutical Services offers development support services, including product release testing. Contact us for more information. -
Product Serialization
Avara Pharmaceutical Services offers primary and secondary packaging services, including serialization to international requirements. Contact us for more information. -
Product Specialization in Hazardous and Complex Chemistry
Avara Pharmaceutical Services offers cGMP production of APIs, with specializations in hazardous and complex chemistry. Contact us for more information. -
Product Spray Drying Solution
Avara Pharmaceutical Services offers clinical-scale drug product manufacturing services, including spray drying. Contact us for more information. -
Product Stability Storage and Testing Service
Avara Pharmaceutical Services offers development support services, including stability storage and testing. Contact us for more information. -
Product Sterile Processing
Avara Pharmaceutical Services offers liquid and lyophilized fill-finish of sterile injectable products from state-of-the-art facilities in North America and Europe. Contact us for more information. -
Product Tablet Manufacturing and Capsule Filling Solution
Avara Pharmaceutical Services offers clinical-scale drug product manufacturing services, including tablet manufacturing and capsule filling. Contact us for more information. -
Product Sterile Manufacturing
Avara Pharmaceutical Services offers liquid and lyophilized fill-finish of sterile injectable products from state-of-the-art facilities in North America and Europe. Contact us for more information. -
Product Milling & Blending of APIs
Avara Pharmaceutical Services offers cGMP production of APIs, including milling & blending. Contact us for more information. -
Product Analytical Methods Development
Avara Pharmaceutical Services offers development support services including analytical methods development. Contact us for more information. -
Product Blending Solution
Avara Pharmaceutical Services offers clinical-scale drug product manufacturing services, including blending. Contact us for more information. -
Product Blister Packaging Solution
Avara Pharmaceutical Services offers blister packaging solutions. We produce 100–300 blisters per minute; form, fill and seal, automated cartoning and inspection; 150 cartons per minute, laser printing, and automated inspection. Contact us for more information.
Frequently Viewed Together
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







































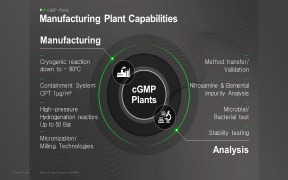

















-comp246698.jpg)