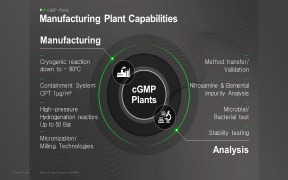
Product Release Testing Service

Product Description
Avara Pharmaceutical Services
-
US
-
2017On CPHI since
-
2Certificates
-
500 - 999Employees
Company types
Primary activities
Categories
Specifications
Avara Pharmaceutical Services
-
US
-
2017On CPHI since
-
2Certificates
-
500 - 999Employees
Company types
Primary activities
More Products from Avara Pharmaceutical Services (26)
-
Product Large-Scale Production (1m3 to 10m3 Reactor Capacity)
Avara Pharmaceutical Services offers large-scale cGMP production of APIs, with 1m3 to 10m3 reactor capacity. Contact us for more information. -
Product Milling Solution
Avara Pharmaceutical Services offers clinical-scale drug product manufacturing services, including milling. Contact us for more information. -
Product Multi-Purpose Equipment and Facility
Avara Pharmaceutical Services offers cGMP production of APIs, with multi-purpose equipment and facilities. Contact us for more information. -
Product Optimized Small-Scale Production
Avara Pharmaceutical Services offers cGMP production of APIs, including optimized small-scale production. Contact us for more information. -
Product Process Development / Optimization
Avara Pharmaceutical Services offers development support services, including process development / optimization. Contact us for more information. -
Product Process Scale-Up & Optimization
Avara Pharmaceutical Services offers cGMP production of APIs, including process scale-up & optimization. Contact us for more information. -
Product Serialization
Avara Pharmaceutical Services offers primary and secondary packaging services, including serialization to international requirements. Contact us for more information. -
Product Specialization in Hazardous and Complex Chemistry
Avara Pharmaceutical Services offers cGMP production of APIs, with specializations in hazardous and complex chemistry. Contact us for more information. -
Product Spray Drying Solution
Avara Pharmaceutical Services offers clinical-scale drug product manufacturing services, including spray drying. Contact us for more information. -
Product Stability Storage and Testing Service
Avara Pharmaceutical Services offers development support services, including stability storage and testing. Contact us for more information. -
Product Sterile Processing
Avara Pharmaceutical Services offers liquid and lyophilized fill-finish of sterile injectable products from state-of-the-art facilities in North America and Europe. Contact us for more information. -
Product Transfer Methods Service
Avara Pharmaceutical Services offers development support services, including transfer methods. Contact us for more information.
Frequently Viewed Together
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance



































































-comp246698.jpg)