Sympfiny® acceptability study among young children with positive results
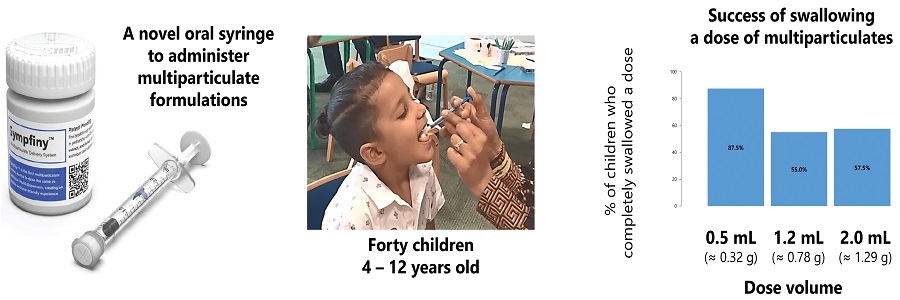
Scientists at the University of Birmingham, UK have evaluated Sympfiny® as device for orally dispensing multiparticulate drugs to children aged 4-12.
Sympfiny® -
developed in collaboration by Röchling Medical and HS Design - is an innovative
system for dosing and delivering multiparticulate, dry powder, and microsphere
drug formulations for oral delivery.
Scientists
at the University of Birmingham, UK have now evaluated Sympfiny® as device for
orally dispensing multiparticulate drugs to children aged 4-12 - with positive
results.
The
study published in „Pharmaceutics“ in August 2020 confirmed that with Sympfiny®,
taste-masked, multiparticulate drugs can be administered to young children conveniently
directly to the mouth without the need for using a co-administration vehicle
such as food or drink.
Sympfiny
was found particularly suitable for a lower dose range (0.1ml).
Read
the full article here: https://www.mdpi.com/1999-4923/12/9/806

Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance