Semaglutide is a new 2nd Generation GLP-1RA drug developed by Novo Nordisk. In Dec. 2019, Semaglutide injection (brand name: Ozempic, 4mg/3ml) was approved by FDA to improve glycemic control in adults with type 2 diabetes. In Jun. 2021, Semaglutide injection (brand name: Wegovy, 2.4mg/dose) was approved by FDA to treat obesity (BMI≥30kg/m2) and overweight (BMI≥27kg/m2).
In Sep. 2019, oral Semaglutide (brand name: Rybelsus, 3mg, 7mg and 14mg/ tablet) was approved by FDA for the treatment of type 2 diabetes. Meanwhile, Rybelsus 25mg and 50mg strengths showed clear effects in Phase II and Phase III clinical trials for weight loss or non-alcoholic fatty liver (NASH).
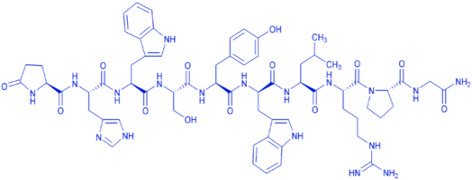
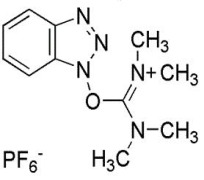
Semaglutide is composed of 31 amino acids and has 94% homology with human GLP-1 (7-37) [Aib replaces Ala at position 8, and Arg replaces Lys at position 34]. In the production of the original drug, the 29-peptide intermediate is fermented by yeast recombinant expression, after which the Lys at position 26 is modified by stearic acid (C18). Then the modified product goes through condensation reaction with His-Aib dipeptide containing protecting group, followed by purification and lyophilization to obtain the Semaglutide drug substance (API).
Peg-Bio’s Semaglutide intermediate (P29, also GLP-1 K34R (9-37)) is a 29 amino acid peptide developed via TE-PEP® recombinant E. coli expression. (TE-PEP® is one of Peg-Bio’s proprietary technology platforms, featuring high efficiency and high-density tandem expression of peptides.) The molecular structure of P29 is C142H216N38O45 and its CAS no. is 1169630-82-3, with a molecular weight of 3175.50 Da. Through fatty acid chain modification and condensation reaction with His-Aib dipeptide, P29 can be used to produce Semaglutide API – and the process route is consistent with that of the original drug’s semi-synthetic processes.






















-comp304625.png)