Pall launches pyrofree sterile, ready-to-use glass vials
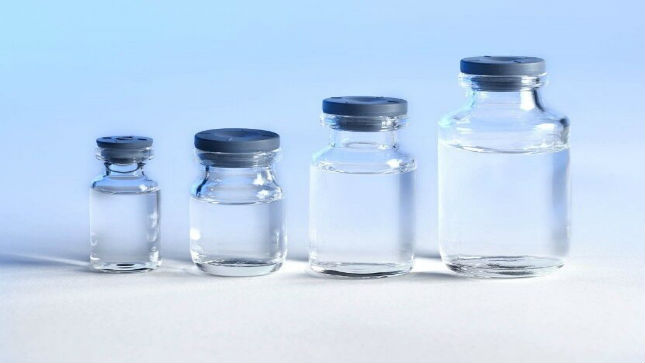
The packaging has been specifically designed for the processing of small batches such as clinical and technical lots, orphan drugs or personalized medicines.
Pall Life Sciences’ is formally launching its Pyrofree glass vial line for sterile, flexible filling from 2 mL to 20 mL at PDA.
The Pyrofree vials product range is a unique solution providing filling processes with ready-to-use glass vials depyrogenated and sterilized according to industrial operations methods applied in vaccines, biotechnology, and pharmaceutical industries, and in hospitals. The packaging has been specifically designed for the processing of small batches such as clinical and technical lots, orphan drugs or personalized medicines. The Pyrofree vials product range offers a unique opportunity to simplify, facilitate and accelerate the filling of these products.
A pyrogen is defined as any substance that can cause a fever, such as endotoxins. The injection of endotoxins into the blood can cause severe hazard to a patient and in worst case, lead to septic shock. Depyrogenation, or the removal of pyrogens, is therefore a crucial need in the aseptic filling process of parenteral drugs. Primary packaging of injectable drugs is particularly controlled for the absence of pyrogens and any chemical agent which may alter drug substances properties and stability. In this case, sterilization of primary packaging (e.g., by ethylene oxide sterilization) represents a risk of introducing chemical agents into drug substances.
The Pyrofree vials product range addresses these needs thanks to its unique packaging technology, allowing sterilization and depyrogenation of the vials by dry heat in their primary packaging under vacuum. The Pyrofree process results in ready-to-use vials which can be used for the filling of injectable drugs without using washing and depyrogenation equipment from the filling line.
The Pyrofree vials product range consists of Type I clear glass vials of 2 mL, 5 mL, 10 mL and 20 mL, supplied with grey stoppers of 13 mm and 20 mm diameters, made of bromobutyl with Flurotec coating.
Related News
-
News CPHI Online Trend Report – 2025 Pharma Packaging Prospects
The pharmaceutical packaging market has seen marked shifts in innovations, priorities, and focus over the last few years. With the rise of hit drugs like GLP-1 agonists and biologics requiring specialised packaging considerations, the strain of meeting... -
News Google-backed start-up raises US$600 million to support AI drug discovery and design
London-based Isomorphic Labs, an AI-driven drug design and development start-up backed by Google’s AI research lab DeepMind, has raised US$600 million in its first external funding round by Thrive Capital. The funding will provide further power t... -
News AstraZeneca to invest US$2.5 billion in Beijing R&D centre
Amid investigations of former AstraZeneca China head Leon Wang in 2024, AstraZeneca have outlined plans to establish its sixth global strategic R&D centre in China. Their aim is to further advance life sciences in China with major research and manufact... -
News US FDA adds haemodialysis bloodlines to devices shortage list
On March 14, 2025, the US FDA published an open letter to healthcare providers citing continuing supply disruptions of haemodialysis bloodlines, an essential component of dialysis machines. -
News Women in Pharma: Manufacturing personal and team success
Our monthly Women in Pharma series highlights the influential lives and works of impactful women working across the pharmaceutical industry, and how the industry can work towards making the healthcare industry and workplace more equitable and inclusive... -
News Pfizer may shift production back to US under Trump pharma tariffs
At the 45th TD Cowen annual healthcare conference in Boston, USA, Pfizer CEO Albert Bourla outlined the potential for Pfizer to shift its overseas drug manufacturing back to the US as pharmaceutical industry players weigh their options against Presiden... -
News Experimental drug for managing aortic valve stenosis shows promise
The new small molecule drug ataciguat is garnering attention for its potential to manage aortic valve stenosis, which may prevent the need for surgery and significantly improve patient experience. -
News Women in Pharma: Connecting accessible pharma packaging to patients – a Pharmapack Special
Throughout our Women in Pharma series, we aim to highlight how CPHI events encourage discussions around diversity, equity, and inclusion initiatives in the pharmaceutical industry.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance
.png)