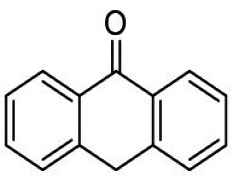
Biocompatibility of Medical Devices according to ISO 10993-1 series
Product Description
galileo research

-
IT
-
2023On CPHI since
-
2Certificates
-
1 - 24Employees
Company types
Primary activities
Categories
Specifications
galileo research

-
IT
-
2023On CPHI since
-
2Certificates
-
1 - 24Employees
Company types
Primary activities
More Products from galileo research (4)
-
Product Consultancy for drug/MD/food development
Our expertise offered in regulatory submissions and knowledgeable strategic skills in planning non-clinical testing programmes can reduce attrition rate and speed up the process.
-
Product Drug efficacy, In vitro and in vivo studies
We offer a full range of studies and bioanalytical services, providing powerful tools and rigorous solutions for preclinical drug development of therapeutic candidates, advance lead compounds towards first-in- human trials, and support for ongoing clinical development -
Product Drug safety, in vitro and In vivo studies
In support of human clinical trials and marketing authorization approval, Galileo Research offers a full battery of toxicity studies for the regulatory development of small drugs, as well as biologics and advanced therapies, in compliance with Good Laboratory Practice (GLP), ICH and OECD guidelines.
-
Product Food safety evaluation
Potential benefits and safety of dietary supplements need to be assessed by scientifically validated means, in the context of Regulation (EC) 2015/2283 on novel foods. Toxicological data, including genotoxicity, information on absorption, distribution, metabolism and excretion (ADME) need to be collecte...
galileo research resources (2)
-
Brochure Galileo Research brochure
Brief description of the company profile and the main services offered by Galileo Research. -
Brochure Galileo-Research-brochure-PDF.pdf
Brief description of the company profile and the main services offered by Galileo Research.
Frequently Viewed Together
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance





















































































%20(2).png)