Biopharma News
Biopharma news
-
News The talk of the show?
Outsourcing, biologics, generics and patient compliance look set to get tongues wagging at this year's CPHI WW. -
News Sartorius Stedim Biotech launches new Sartocheck 5 Plus filter tester
Highest level of data integrity as requested by regulatory authorities. -
News Sanofi opens its first digitally-enabled, continuous manufacturing facility
One of the first digital manufacturing facilities in the world to use continuous, intensified biologics production technology. -
News Discover Roquette’s latest innovations and our new Biopharma Innovation Center!
Roquette works closely with customers in the development of innovative pharmaceutical formulations and new biopharma solutions. -
News Industrial-scale manufacture of therapeutic exosomes
Coordinated purification process development service accelerates progress to clinical trials, and scale-up manufacturing. -
News Data on lead ADC demonstrates effective tumour regression
IKS01 shows marked anti-tumour efficacy in pre-clinical models of ovarian and lung cancer. -
News Arctoris secures funding to advance novel robotic drug discovery platform
Platform will enable scientists and biotechnology entrepreneurs worldwide to make discoveries faster and more efficiently, -
News GSK invests $120 million in next-generation US biopharma manufacturing facility
Part of nearly $400 million in US manufacturing investments to deliver on the company’s pipeline and bring medicines to patients faster. -
News FUJIFILM Irvine Scientific launches BalanCD Gal Supplement for biotherapeutic development
Delivers enhanced galactosylation for improved protein quality, and antibody binding and function. -
News Sandoz enters global deal to commercialise proposed biosimilar natalizumab
Worldwide agreement with Polpharma gives Sandoz commercialization rights to proposed biosimilar natalizumab for relapsing-remitting multiple sclerosis. -
News Rapid adoption of Carterra’s LSA instrument drives European expansion
Company's antibody screening platform minimizes the risk of missing a blockbuster. -
News HALIX starts operational production with new cGMP facility in Q4-2019
The five-level production facility contains a state-of-the-art manufacturing line for viral vaccines and viral vectors and a separate protein manufacturing area with a capacity up to 1,000-L single-use bioreactors. -
News Delivering complete aseptic vial handling solutions
Bringing freeze drying and vial washing/sterilizing technologies together with advanced and comprehensive vial and syringe filling capabilities. -
News Sartorius launches new services for mammalian cell bank manufacturing
Integrated package of new and established services saves time and minimizes risks. -
News Needle free: Erase wrinkles without injection
Meet REVOLUTION BEYOND INNOVATION at Cphi Worldwide 2019, Booth No. 42C06 -
News New state-of-the-art cleanroom boosts production of anticancer drugs
The GMP facility has been specifically designed to deliver the next generation of anticancer blockbuster ADC drugs for patients globally. -
News Eisai collaborates with University of Dundee on cancer drug discovery
It is hoped that research into PROTACs will lead to new drug discoveries for proteins present in cancer, which are difficult to treat with conventional small molecule inhibitors. -
News Fujifilm Irvine Scientific to open new cell culture media manufacturing site in Europe
The space will support cGMP manufacturing of animal component-free, dry powder media, liquid media, and downstream bioprocessing liquids. -
News Fresh data on protein interactions in Alzheimer’s disease
Demonstrates the unique ability of diffusional sizing to assess protein binding in-solution and for difficult-to-study systems. -
News Cyclolab Ltd strengthens its position in the market
Cyclolab Ltd. is an R&D and cGMP manufacturing company focusing on all aspects of cyclodextrin science, manufacturing and applications for cyclodextrin, Dexolve, Sugammadex, formulation development, custom synthesis, Niemann Pick C. -
News New ambr 250 modular bioreactor vessel for cell and gene therapy applications
Designed for gentle stirring and optimum growth of cell lines. -
News New-generation incubator brings the latest technology to mid-capacity microplate cell culture
New Thermo Scientific Cytomat 2 C-LiN Series Automated Incubator delivers robust and reliable culture solution for cell-based pharmaceutical applications. -
News AbbVie to acquire Allergan in transformative move
Transaction will create a leading biopharmaceutical company with approximately $48 billion in combined 2019 revenue. -
News HCP Kit for automated impurity analysis of biotherapeutics
Ready-to-use immunoassay kit increases analytical output and productivity in bioprocess workflows. -
News Inaugural Bio Integrates conference highlights industry's inefficiency in developing products
Industry leaders give voice to issues and trends shaping the biotech sector, including the importance of collaboration. -
News Affordable measles and rubella vaccines using NevoLine manufacturing platform
Univercells' propriety bioproduction platform has already proven success. -
News Sphere Fluidics secures funding to support its single cell analysis system
Funding will also be used to further expand operations in the UK and US. -
News CPHI China opens with analysis pointing to a surge in growth in 2019
Regulatory reforms and harmonisation reported as key drivers of industry in China. -
News Catalent completes acquisition of gene therapy leader
Acquisition of Paragon Bioservices brings specialized expertise in adeno-associated virus and will enhance Catalent’s biologics business and end-to-end integrated biopharmaceutical solutions for customers. -
News Vibalogics increases capacity with a new 200-L line for virus manufacturing
Company has also invested in an Äkta-ready chromatography system for downstream processing and liquid handling equipment to deal with increased volumes. -
News New cell culture microbioreactor system improves clone selection
SSB's new generation of ambr 15 supports cell and gene therapy applications, including HEK293 for viral vector production, T-cells, iPSCs and other immune-derived cell lines. -
News Surge of Indian biosimilars market forecast in 2019
India predicted to be one of the world’s ‘fastest growing bio’ hubs in 2019, fuelled by new biosimilars production. -
News Isotype-specific secondary antibodies for improved signal detection
Offers an alternative to cross-adsorbed secondary antibodies when absolute specificity is required. -
News New China-based biotechs fueling growth in manufacturing across China
New guidelines will see poorer quality manufacturers drop out of the market. -
News 'Drugs from bugs' joint venture
Lonza and Chr. Hansen create a strategic joint venture to become the partner of choice for developing and manufacturing live biotherapeutic products for pharma and biotech customers. -
News ChargePoint Technology launches single-use technology as part of powder transfer solutions portfolio
Company's hybrid solution claims superior performance compared with tradition single-use technologies. -
News Sartorius integrates online biomass measurement to its ambr systems for microbial applications
Scientists to rapidly obtain detailed process understanding and control over their microbial cultures. -
News Bringing next generation single-use sensor technologies to the life science market
Pall and Broadley-James’ combined expertise proves complementary in addressing critical customer pain points in modern bioprocessing. -
News Novel systems for membrane chromatography
Optimally run membrane chromatography processes will provide higher productivity, smaller-scale operations and increased robustness. -
News Biogen acquisition boosts its ophthalmology pipeline
Nightstar Therapeutics has two potentially first-in-class mid- to late-stage clinical assets as well as preclinical programs. -
News Japan solid dose market predicted to bloom in 2019
Executives predict Japan to be the second fastest growing mature market for solid dose drugs in 2019, and the second fastest overall country for biologicals growth. -
News Partnership expands to develop a new treatment for pachyonychia congenita
MedPharm and Palvella Therapeutics hope to develop the first approved treatment for PC in the US and Europe. -
News Andrew Alliance and Sartorius collaborate to provide software-connected pipettes for life science research
Researchers to benefit from an innovative software-connected pipetting system, bringing improved reproducibility and traceability of experiments to life-science laboratories. -
News Hitachi Chemical to acquire apceth Biopharma
Acquisition will strengthen Hitachi's presence in the second-largest cell and gene therapy market in the world. -
News Sartorius joins the community of the National Institute for Innovation in Manufacturing Biopharmaceuticals
Company hopes to contribute substantially to further optimizing and accelerating the current manufacturing processes in the biopharmaceutical industry. -
News Particle Sciences partners with Encube Ethicals to develop novel vaginal rings
The partnership will focus on rings for the administration of existing approved drugs, improving drug effectiveness and patient compliance. -
News SGS expands testing capabilities at its Glasgow facility
Clients will benefit from a fully comprehensive range of validated biosafety methods to support cell bank and viral vaccine manufacturing and lot release of drug product. -
News TC BioPharm creates allogeneic cell banks for CAR-T cancer therapy products
The first completed bio-banks will be used to develop more cost-effective, safe and efficacious cancer treatments. -
News Alvotech completes US $300 million financing deal
Fuels growth and biosimilar development in a fast-growing market. -
News Sartorius introduces BIOSTAT RM TX single-use bioreactor
Flexsafe RM TX bags enable reliable process performance for optimal cell growth. -
News Univercells introduces breakthrough vaccine manufacturing platform
The automated NevoLine bioproduction system that facilitates safer, faster and closed bioprocessing in a much smaller footprint. -
News Allergan to establish R&D presence in Cambridge, Massachusetts
The new, strategic presence will allow the company to more easily interact and engage with venture firms and start ups in the area. -
News Expedeon signs supply and license agreement for is Lightning-Link technology
The technology will enable Cell Guidance Systems to manufacture TRIFic detection assays on-demand with highly reproducible and scalable results. -
News BMS and Celgene merge to create premier innovative biopharma company
Significantly expands Phase III assets with six expected near-term product launches, representing greater than $15 billion in revenue potential. -
News Pall opens Center of Excellence in Shanghai
The CoE showcases the latest end-to-end technological solutions for biomanufacturing. -
News New gene therapy manufacturing facility for Orchard Therapeutics
Enhances company's capacity to develop and deliver lentiviral vector and gene-corrected hematopoetic stem cells for wide range of diseases on a global scale. -
News Grant winners to investigate continuous manufacturing for gene therapies
Cobra Biologics, Pall, and the Cell and Gene Therapy Catapult share £1.5 million grant from the UK's innovation agency. -
News ADC Bio secures additional equity round investment
Extra funding for strategic move into downstream formulation and fill finish capabilities and full exploitation of Lock-Release as new ADC manufacturing paradigm. -
News SSB and Lonza modify relationship for supply of cell culture media
Sartorius Stedim Biotech will continue to offer Lonza media and buffer products but under a non-exclusive agreement. -
News bioLIVE to merge with BioProduction Congress for 2019 event
BioProduction’s content platform will be added to the bioLIVE exhibition, creating one of the world’s largest event hubs for the bio industry. -
News Sandoz pulls the plug on US biosimilar rituximab
Company will focus on robust biosimilar portfolio for unmet access and sustainability needs. -
News Three-way collaboration to advance gene therapy
Partnership aims to increase the robustness and reduce costs for the manufacturing of AAV vectors. -
News Bioconjugation expertise and increased productivity in bio over next 5 years
Smaller bioreactors and high-density perfusion techniques needed as manufacturing makes significant efficiency improvements. -
News Alvotech receives manufacturing licence for its biopharmaceutical facility
Facility to focus on developing and manufacturing the company's biosimilar portfolio. -
News Complete product lifecycle management in one location
Lonza expansion includes drug substance development, and drug substance and drug product manufacturing. -
News BIA Separations expands with new upstream processing facility
Provides single source of expertise for scaled production of complex biologics for gene therapy and vaccines. -
News Partnership to deliver integrated, off-the-shelf biosimilar manufacturing solutions
Pall and Aetos to advance the market impact of biosimilars and deliver lower-priced, high-quality options to end users. -
News New SGS biopharmaceutical testing capabilities in Illinois
The new services will focus on quality control analysis and stability testing of biopharmaceuticals. -
News Novartis to divest the Sandoz US dermatology business and generic US oral solids portfolio
The Sandoz US portfolios to be sold to Aurobindo include approximately 300 products, as well as additional development projects. -
News Sartorius and Repligen partner to introduce next-generation perfusion-enabled bioreactors
Customers to benefit from simplified, scalable solutions for intensified cell culture. -
News Plasticell wins military contract to develop regenerative medicines for the battlefield
Company to use its combinatorial stem cell screening platform to develop technologies for the conversion of pluripotent stem cells into platelets. -
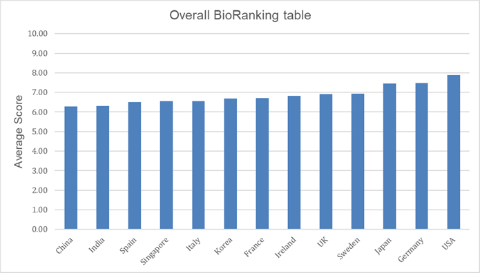
News Bio processing and manufacturing league table ranks US top and China bottom
US, Germany and Japan revealed as tier one nations in all categories with Sweden ranking the best of the rest. -
News New biopharmaceutical testing laboratory to open in Geneva
SGS's expansion will enable the service provider to offer a full ICH Q6B physico-chemical characterization of biological products. -
News Exosome isolation manufacturing and characterization
New service to reliably and reproducibly isolate exosomes from almost any biofluid. -
News Oxford Genetics signs major supply and licensing agreement for CRISPR engineered mammalian cell lines
Company moves away from manual processing in favour of automated, scalable platforms. -
News Extended distribution agreement enables customers to continuously improve their bioprocesses
Agreement includes Pall's next-generation Kaneka KanCapA 3G sorbent for the primary capture of mAbs from clarified cell culture. -
News Amgen breaks ground on next-generation biomanufacturing plant in Rhode Island
The plant will be first-of-its-kind in the US. -
News Partnership struck to aid bio manufacturers
Pall and BioSciencesCorp unite to provide "total solutions" for today's drug manufacturers. -
News WuXi STA and Antengene sign development and manufacturing agreement
WuXi STA chosen for its end-to-end CMC platform for new drug development. -
News New screening company exploits the latest advances in microfluidics and 3D culture
The ability to micro size drug-cell interactions will allow pharmaceutical and biotech companies to do 100x more testing for the same money spent as well as increasing productivity. -
News South Korea's finished drug market growth driven by generics
FDF launches at CPHI Korea as country set to become regional hub. -
News Plasticell leads gene therapy manufacturing consortium
Consortium to develop advanced technologies for the manufacturing of ex vivo gene therapies. -
News Ingredients to improve bioprocess manufacturing efficiency
Experts warn bio industry needs to do more to create a sustainable pipeline of talent. -
News CellGenix completes first step of facility expansion
Next expansion step will increase finished product capacity for recombinant proteins by more than tenfold. -
News Extended co-operation agreement to benefit Sartorius customers
Agreement will enable customers to achieve easier scalability of processes ranging from lab-scale to commercial-scale production of biopharmaceuticals. -
News Thermo invests $50 million to enhance biologics capabilities
With the capacity expansion, Thermo Fisher will have the second largest base of single-use bioproduction capacity at a CDMO in the world. -
News Cost-effective, scalable process development of vaccines in cell culture
Sartorius Stedim Biotech introduces its mini microcarrier bioreactor for culturing adherent cells. -
News Wacker acquisition increases cost-effective pharmaceutical production
The contract manufacturer strengthens its pharmaceutical-protein business with Dutch acquisition -
News Amgen to build first-of-its-kind biomanufacturing plant in the US
Revolutionary, innovative plant offers greater flexibility, speed and efficiency. -
News JXTGE to sell Irvine Scientific to FujiFilm
With the acquisition of Irvine Scientific, FujiFilm will have a full product line supporting customers supplying antibody drugs to assisted reproductive technologies and cell therapy. -
News bioLIVE to introduce global biopharma country ranking
Global analysis will rank each major biomanufacturing country’s market growth potential, innovation and competitiveness. -
News Top marks for Cherwell’s Redipor prepared microbial media products
Extensive range of ready-to-use culture media available in bottles and plates. -
News ProJect Pharmaceutics expands facilities for formulation and lyophilization process development
Expansions to meet safety requirements for GMO S2 and BSL-2 biological products. -
News Revolutionary downstream bioconjugation method under development
New manufacturing paradigm will save 25% of total costs and the need for proteinaceous A & G resins. -
News A powerful new tool for the separation of charged species
A new analytical tool, Electrical Asymmetrical Flow Field-Flow Fractionation (EAF4) is showing much promise in biopharmaceutical and nanoparticle applications. -
News New Pall business unit launches to focus on end-to-end drug manufacturing models
Pall Biotech to focus on enabling drug manufacturers to rapidly transition from pre-clinical to commercial manufacturing. -
News Ablynx - Sanofi's new acquisition
Strengthens Sanofi's R&D strategy with innovative Nanobody technology platform. -
News Sandoz and Biocon to collaborate on next-generation biosimilars
Collaboration will leverage combined strengths of development, manufacturing and commercialization of biosimilars. -
News SSB to equip Abzena's US-based development and manufacturing sites
SSB will provide both facilities with end-to-end process solutions in single-use format. -
News Momenta and Mylan to development proposed biosimilar to Eylea
Targeting the initiation of a pivotal patient clinical trial in the first half of 2018. -
News ANVISA approves Biocon and Mylan's biosimilar Trastuzumab
Libbs will commercialize the product in Brazil under the brand name Zedora. -
News Major changes for bio supply forecast in 2018 by bioLIVE
Integration of supply chain a critical issue as industry globalises, with a resurgence in European manufacturing and biotech markets forecast. -
News Hikma signs licensing agreement with Celltrion for third biosimilar product in MENA region
Truxima is mAb biosimilar to Roche's MabThera (rituximab). -
News CMC Biologics and Harpoon Therapeutics collaborate on exciting new TriTAC molecules
First clinical candidate, HPN424, is in development for the treatment of metastatic prostate cancer and expected to enter Phase I clinical trials in 2018. -
News SGS introduces GMP DNA sequencing service at its Glasgow, UK, laboratory
Service complements existing services for this testing, to provide comprehensive analysis and characterization solutions for clients involved in the production and manufacturing of biopharmaceutical products. -
News SGS launches new large molecule bioanalytical services
Investment builds on company's expertise in small molecule analysis and the use of LC/MS-MS techniques. -
News Expansion of CSL Biotech facility drives advanced manufacturing growth
The development will help meet growing global demand for albumin. -
News Global ADC Bio clients to benefit from new dedicated bioconjugation facility
The design, planning and building work commences immediately and will deliver a purpose-built, dual-stream facility. -
News New clarification platform delivers robust, accelerated performance across multiple mAb processes
The Stax mAx, a single-use harvesting platform, minimizes the impact of process variability between batches. -
News Poor aggregation will see ADC targets fail or face long delays
Experts at ADC Bio warn of impending problems in the ADC pipeline with millions wasted in development costs. -
News Celonic acquires Glycotope’s biomanufacturing facility
Combined technology assets will provides pharmaceutical and biotech customers the unique opportunity to select a tailored expression system. -
News Eurofins creates one of the largest dedicated testing sites of its kind in the UK
£4 million investment will allow the Biopharmaceutical Product Testing business to expand its finished product and raw materials testing and increase capacity to deal with higher volumes. -
News New dedicated biopharma event launches alongside CPHI Worldwide 2018 in Madrid
New event to create unique synergies between the parallel worlds of large, small molecules and contract services. -
News Batavia expands its viral vector and cleanroom facilities to stay on top of demand
Investments will help the company to bring candidate biopharmaceuticals from bench to clinic. -
News Construction starts for Sartorius' new Cell Culture Technology Centre
The 30-million-euro investment will accommodate the growing needs of customers. -
News PnuVax awarded $29.4 million grant to advance innovative vaccine into clinical trials
Use of PnuVax’s unique patented conjugation platform technology to produce the vaccine is expected to speed up biomanufacturing and increase yield for a greatly reduced per-dose cost. -
News 3SBio accelerates expansion of its global biologics platform
The Chinese biopharmaceutical company acquires the Canadian biomanufacturing business of Therapure. -
News Bayer submits BLA in the for BAY94-9027 – a long-acting factor VIII for the treatment of hemophilia A
Pivotal studies with BAY94-9027 showed that bleed protection was achieved with extended dosing intervals. -
News Merck successfully closes biosimilars transaction
Step towards strategic alignment of R&D resources to Healthcare priorities. -
News Biogen’s adalimumab biosimilar referencing Humira, is approved in the EU
Biogen becomes the first company with approved biosimilars for the three most prescribed anti-TNF biologic treatments in Europe.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance










.jpg)
.jpg)





.jpg)






.jpg)
.png)



%20(2).jpg)
%20(2).jpg)


.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
%20(1).jpg)









.jpg)

































.jpg)
.png)









.jpg)
.png)








