Cayman Pharma s.r.o
About Cayman Pharma s.r.o
Certifications
Categories

-
CZ
-
2015On CPHI since
-
3Certificates
-
100 - 249Employees
Company types
Primary activities
Meet us at
CPHI Milan 2024
Fiera Milano, Italy
08 Oct 2024 - 10 Oct 2024
Products from Cayman Pharma s.r.o (5)
-
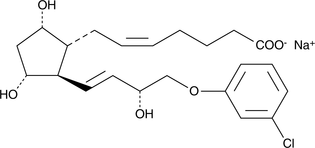
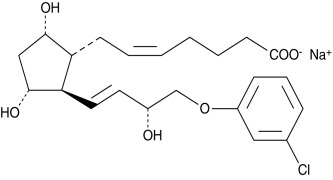
Product CGMP (+)-Cloprostenol (sodium salt)
(+)-Cloprostenol is the active enantiomer of Cloprostenol. Cloprostenol is used in veterinary medicine as a luteolytic agent for the induction of estrus and the treatment of reproductive disorders in cattle, swine, and horses. The ASMF for (+)-Cloprostenol (sodium salt) is on file in several EU member states. -
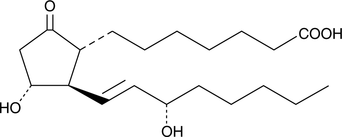
Product CGMP Alprostadil
Alprostadil is a vasodilator, and finds clinical use in treating erectile dysfunction, in improving blood flow in peripheral vascular disease, and in maintaining coronary circulation in neonates with vascular anomalies. The DMF for Alprostadil is on file with the US FDA, Canada, India, in many EU member st... -
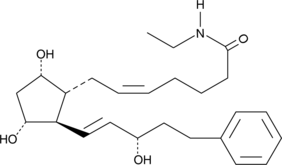
Product CGMP Bimatoprost
Bimatoprost is a potent FP receptor agonist that finds clinical use as an ocular hypotensive agent for the treatment of glaucoma. The DMF is on file with the US FDA, Canada, and in several EU member states. -
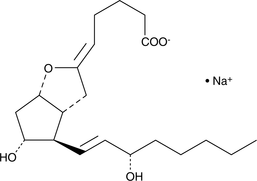
Product CGMP Epoprostenol (sodium salt)
Epoprostenol is a potent vasodilator and antiplatelet substance with a very short physiologic half-life. It is used for the treatment of pulmonary hypertension. The DMF for Epoprostenol Sodium Salt is on file with the US FDA, Canada, Japan, Australia, and several EU member states. -
Product CGMP (±)-Cloprostenol (sodium salt)
Cloprostenol is used in veterinary medicine as a luteolytic agent for the induction of estrus and the treatment of reproductive disorders in cattle, swine, and horses. (±)-Cloprostenol sodium salt is manufactured at Cayman Pharma, our Czech subsidiary. GMP material, which conforms to the USP and BVP Monogr...
Cayman Pharma s.r.o Resources (2)
-
News Cayman Chemical Receives CEP for Latanoprost API from EDQM
ANN ARBOR, MI, SEPTEMBER 12, 2023—Cayman Chemical, a world leader in the manufacture of prostaglandins for both research use and as active pharmaceutical ingredients (APIs), has been granted a Certification of Suitability to the monographs of the European Pharmacopoeia (CEP) by the European Directorate for the Quality of Medicines and HealthCare (EDQM) for its latanoprost API, effective August 28th, 2023. Cayman Chemical is the first company in the USA to receive a CEP for latanoprost. -
Brochure API Manufacturing
Learn more about the importance of process understanding and Cayman's commercial API production
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance