CPHI Online is the largest global marketplace in the pharma ingredients industry
-
Products0
-
Companies0
-
Articles0
-
Events0
-
Webinars0
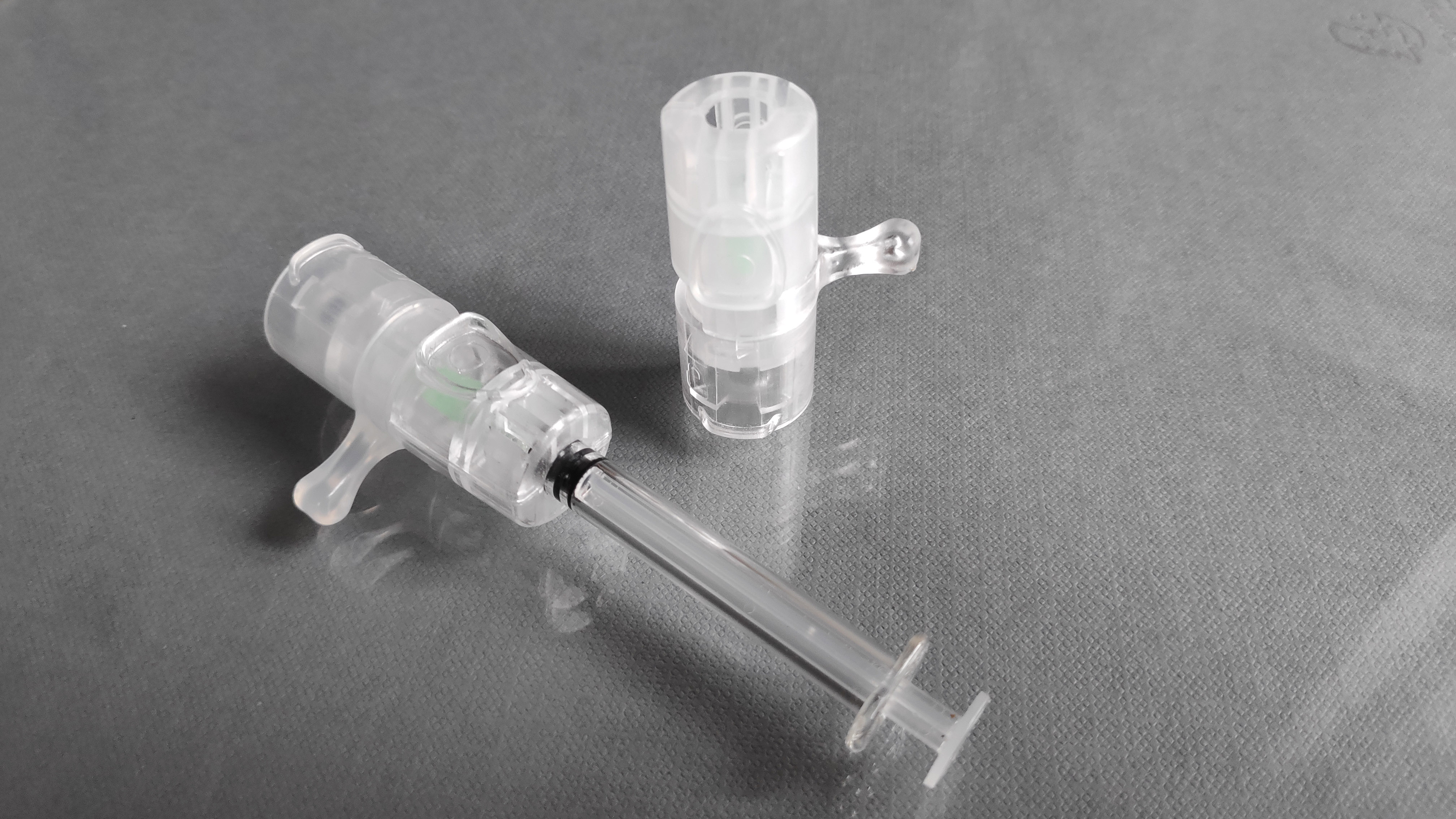
VAX-ID®
Product Description
VAX-ID® is an award-winning patented drug delivery device suited for reliable intradermal injection with high ease of use. The devices are suited for all ages and populations. The intended users are healthcare providers.Devices are currently available for investigational use only (IUO) and research use only (RUO). CE marking and FDA approval are expected early 2025
Idevax

-
BE
-
2022On CPHI since
-
1Certificates
-
1 - 24Employees
Company types
Drug delivery device Supplier/Manufacturer
Primary activities
Medical Devices
Categories
Specifications
- DetailsVAX-ID® can be preconfigured with a 32G, 30G, or 27G needle with a predefined penetration depth of less than 1mm allowing for reliable drug delivery in the dermal layer of the skin. The devices are suited for delivery of small volumes of medicinal substances (typically 0.05cc – 0.2cc per injection).
- Selling PointsConvenience; Immune Health; Developed by a multidisciplinary team, VAX-ID® allows for
- Accurate injection at a predefined penetration depth- Dose sparing opportunities
- Activation protection and needle-stick injury prevention
- Low in pain and no needle phobia
- Easy to use, potentially leading to self-administration
- User independent
Idevax

-
BE
-
2022On CPHI since
-
1Certificates
-
1 - 24Employees
Company types
Drug delivery device Supplier/Manufacturer
Primary activities
Medical Devices
Idevax resources (1)
-
Video How VAX-ID works
The video shows how VAX-ID, a skin drug delivery device for reliable injection in the dermal layer of the skin, works.2025-01-13
Recommended Products
-
Product Medicurtain
• Surgical adhesion barrier • Composition: Sodium hyaluronate 10mg/1mL, HES 5mg/1mL • Indication: To prevent or reduce post surgical adhesion formation after hysteroscopic (gynecology), abdominal, spinal, thyroid and nasal/sinus surgeries. • Dosage and administration: Refer to the brochure f...
-
Product Whitepaper Design for manufacturing
Design for Manufacturing:
Why, how and when?
Minimized risks, increased production speedand a lower cost price. A toolmaker’s Design for Manufacturing (DFM) expertise can have a large impact on your production process. But what is the most effective way to collaborate with a toolmaker? Wh...
-
Product MK™ Fine Mist Sprayer
Deliver precision dosing and superior spray performance for your Rx and OTC medications. Meet MK™—a fine mist sprayer designed to deliver precision dosing and superior spray performance. With its excellent priming, atomization, and clean output, it delivers an effortless spray experience for consumers. At...
-
Product Measuring Devices
Measuring devices like dosing cups and spoons are available in various sizes (5 ml to 30 ml) and materials, primarily PP. With precision-engraved markings and cleanroom production, these devices ensure accurate dosing for liquid medications.
-
Product Pharma Fluid Handling
Raumedic manufactures complex tube sets for fluid handling in single-use, multi-use, and hybrid systems along with pharma tubing. No matter whether you are interested in manual small-batch production or fully automated long-running favorites – we observe strict clean-room...
-
Product One-stop solutions by GEMÜ
GEMÜ is your one-stop shop for customized solutions. Advanced technologies have been integrated to meet the demand for complete solution providers in our markets. We offer our customers processes such as laser welding, printing through to completely assembled, tested, and packaged system solutions, as ...
-
Product INJECTABLES
Requiring injectables.EU-GMP is a must.
EU dossier is a must.
Licensing and supply model or distributorship.
-
Product BrudyAl Plus Multi
BrudyAl Plus Multi is an eye drop for the lubrication of the cornea. It is preservatives free and the effect it is immediate. -
Product SENSOkidCAP®
SENSOPLAST® combines the advantages of tamper-evident closure with a child safety solution. The cap can only be removed from the bottle by simultaneously pressing and turning the cap. At the same time, tearing off the tamper-evident ring reveals the initial opening.
This special design has prove...
-
Product Automatically tracking children's medication
As a busy parent, managing a child's medication can be hard. Team Consulting integrated connectivity into a syringe to help parents keep track of their child’s medication through a smartphone app.
Using a small, low-cost sensor, this connected syringe automatically tracks when and how much drug...
-
Product Dosecare
Dosecare is a dosing syringe for oral liquids.
It differs from the others as it allows you to simply and intuitively save the dose of liquid to be administered. Therefore once the dose is set, dosing errors are prevented, time is saved, there is no waste and soiling.
Thos...
-
Product Sustainable Materials for Medical Devices
Hostaform® MT® POM ECO-B, Celanex® MT® PBT ECO-B, and Hytrel® TPC ECO-B are sustainable drop-in solutions for the reduction of CO2 footprint of your medical device. Chemically identical to their respective conventional materials, they incorporate bio-based feedstock to lower the CO2 footprint via a mass-b...
-
Product Urine catheter products & drainage bag machines
We offer a wide range of solutions for the manufacturing of various urine catheter products; from processing the pre-fabricated catheter, welding the urine receiving bag, to manufacturing and integrating in-bag fluid dispensers into the receptacle, or applying lubricant along the catheter.
...
-
Product Physical & Functional Testing
• Combination Device Testing • Needle-based Injection System • Device Verification & Validation • Bespoke & Custom Testing • Particulate Matter Contamination • Packaging Validation & Testing • ASTM D4169 Transit Testing • ASTM F1980 Accelerated Ageing • ISO 11607 Sterile Barrier Valid...
-
Product Medical devices for gynecological application
INGE produces medical devices for vaginal liquid products, creams and suppositories. 50 ml to 180 ml bottles for vaginal shower made of a soft, easily compressible material, also Green. Bottles are available in various shapes - cylindrical or elliptical - customizable with front and back engraving and one-...
-
Product Medical Devices
Nolato provide the healthcare and pharmaceutical industries with development and manufacturing of complex product systems and components based on advanced polymer technology and automation.
Extensive offering-We are a global medical solutions provider serving the pharmaceutical and healthcare industri...-comp247874.png)
-
Product Collagen Membranes
You need a collagen matrix with particular features? Viscofan BioEngineering develops custom-tailored scaffolds in alternative sizes, shapes, and individual thicknesses (20 – 200 μm) upon request. We adjust permeability, elasticity, degradation time, tensile strength, swelling properties and other characte...
-
Product Dermal Filler | BIO-Rejuvenation | Modified Hyaluronic Acid
SEMICAL® Dermal Filler; Supporting the extracellular matrix, increasing intra-cutaneous volume, is also an injection method used in the treatment of wrinkles caused by cicatrix or skin aging. USAGE AREAS SEMICAL® Dermal Filler for face applies to the following regions: • Nasolabial lines
...
-
Product Customized Folding Carton
Customized Folding Carton with insert pocket for product/medical device.
-
Product Serialisation and track & trace solutions for Healthcare
To protect patients and drug manufacturers from seriously damaging counterfeits, a high level of product and process transparency and authenticity is in place. The industry is relying on serialisation and track & trace – and there are several, different country-specific requirements, that regulate ...
-
Product SINOVIDOL
SINOVIDOL is a Bio-regenerant based on collagen precursors, which facilitate chondrocytes to produce tropocollagen rapidity that polymerizes in the extracellular matrix of the cartilage forming real collagen. Indicated for the treatment of restructuring of the cartilage and its annexes. Is a potent eutrop...
-
Product Stoppers & Caps
Features of our Closures; • Have passed the leakproofness test and are reliable, • Can be supplied with an aluminum seal installed, • Can be produced in different colors, • Our child-proof caps provide the ideal balance between innovative design, ease of use and reliability, ...
-
Product Hygienica Plus lotion against lice and nits
An effective product for removing lice and nits without containing any chemically or biologically active chemicals. It is effective against lice and their nits, it is not toxic. Safe for children over 3 years, pregnant women, lactating women, asthmatics and people with allergies. Dimethicone acts physicall...
-
Product Capsules/tablets
Biolevox™ Neuro (30 film-coated tablets)a set of ingredients that support the functioning of the nervous system.
Biolevox™ Complex bis (90 film-coated tablets) supplements the diet with ingredients found in joint structures thus supports the their functioning.
C...
-
Product Regulatory Affairs
Our team of Regulatory affairs experts make our service offerings stronger.
Following are our Regulatory offerings:
• Regulatory Operations, Affairs
• Regulatory Intelligence, Information Management COTS
• Regulatory Strategy & Business Consulting
• Health Autho...
-
Product D2Mix™ Reconstitution Devices
• Quick and easy preparation with simple handlings • Contamination risk reduction by reducing preparation steps • Less exposure and mis-injection risk by their unique device design • Enhancement of user-safety by the needle-free design
-
Product JOYDROPS LUBRICANT GELS-DELAY GEL
Turkuaz medikal kozmetik dis ticaret ltd provides wide range of products which includes joydrops lubricant gels-delay gel. Contact us for more informations.
-
Product Priiotize Make Ready
Where artwork meets smart work!Make Ready helps you prepare your healthcare packaging artworks for print production. This tool creates formats based on existing approved layouts. Your original PDF will not be changed via our workflow, but will be enhanced with a metadata concept using our proven techn...
-
Product BIEFFE FIBER - medical device
BIEFFE FIBER is a class IIa medical device with a laxative effect containing Fibersol®-2.
PRODUCT
The product is intended for the treatment of the symptoms and the prevention of constipation. BIEFFE FIBER improves symptoms ofconstipation by regularizing the stool frequency, facilitates the ...
-
Product Adhesive First Aid Dressing
Breathable film Superior strong hypoallergenic adhesive. Latex Free.Medicated with superior antibacterial Benzalkonium Chloride 0.5% w/wFirst Aid remedy for small cuts, wounds & abrasions.Available in flesh colour or transparent & food grade blue colourAlso available ...
-
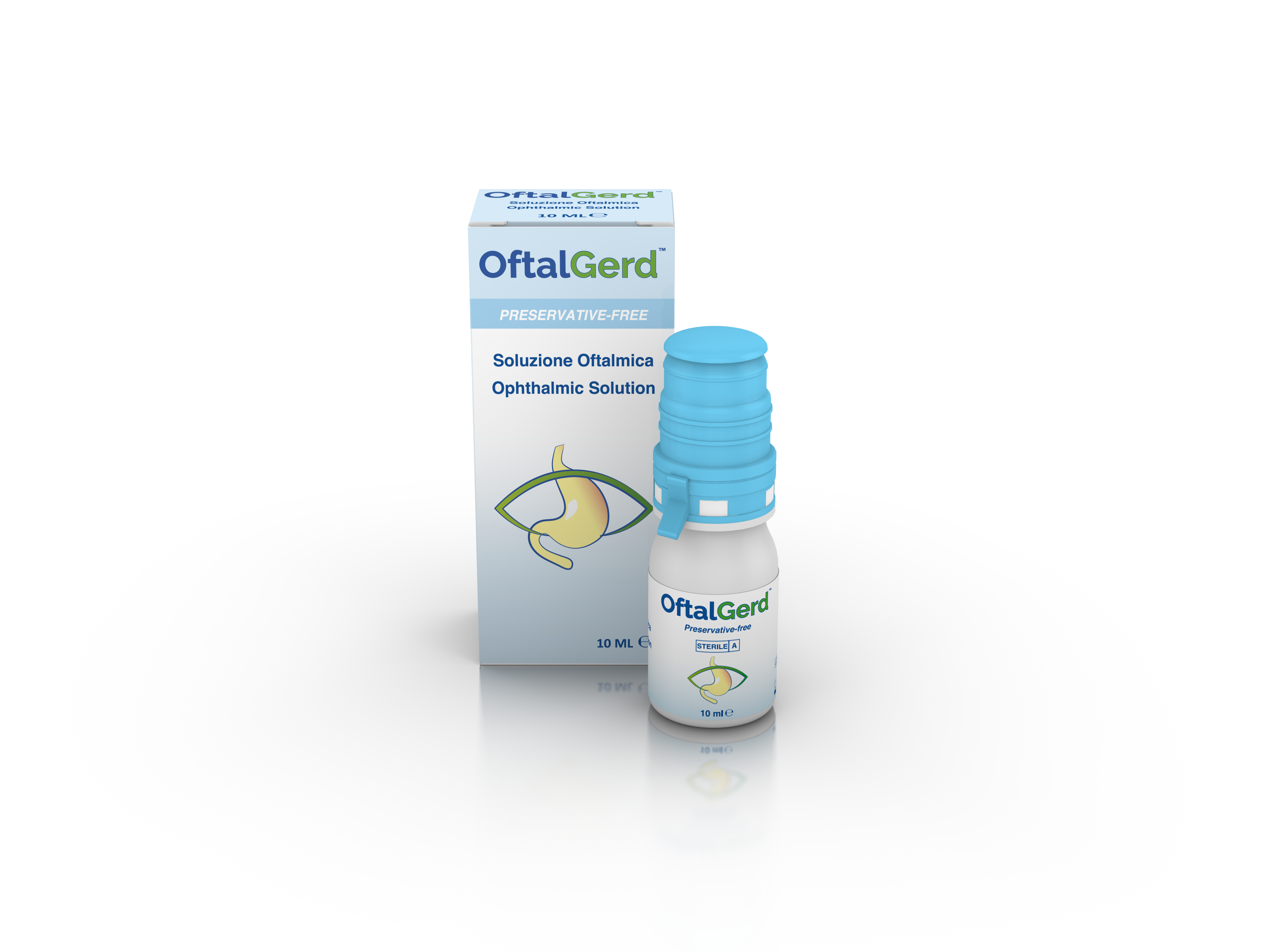
Product OFTALGERD - A special formula for Reflux suffers - Preservative Free Oph...
A special formula for reflux sufferers.
Indications.
Medical Device indicated for the treatment of tear dysfunction symptoms (irritation, itching, tearing, foreign bodysensation, eye fatigue sensation, etc…) either transitory or associated with chronic diseases. OftalGerd Ophthalmic Solution, tha...
-
Product Nutridef Aerosol
Treatment of cold and allergy symptoms.Range of application
• in case of viral and allergic rhinitis
• in case of nasal congestion
• in case of acute and chronic sinusitis
• in case of prevention and treatment of catarrhal otitis.
Complete formulation for the whole family and wide range of i...
-
Product PEBD CONTINUOUS LINER
Infinite bag manufactured in ISO5 cleanroom, of 15 to 40 meters dimensions, to put in insolators, or for waste throwing after productions, for safer working area for workers and no eyes/hand/nose contact with any toxic product. Can be manufactured with L2 special resistivity specifications, to prevent ...
-
Product TIABLO WIPES (OCULAR SURGERY AND BLEPHARITIS, EYELID CLEANING)
TiABLO, ophthalmic gauzes soaked in TiAB solution, provides unbeatable cleaning and protection. TiAB is a complex of Silver, a powerful antiseptic and Titanium, that maintain Silver in its active formula. TiABLO kills common bacteria responsible for ocular infections. It is especially recommended in tr...-comp246544.jpg)
-
Product JUVIAGEL – Vaginal moisturizing gel
Problem
Vaginal dryness (or atrophy) is a condition caused by a decreased level of the oestrogen hormone. It is characterized by a thinning, dehydrated and possibly inflamed vaginal wall. A dry vagina means uncomfortable or painful sex. In more severe cases it may cause a dry, itchy or even b...
-
Product Springs and Stamped & Bent Parts for Pen Injectors
Tailor-made solutions in springs, stamping & bending and assemblies for enhanced pen injector performance and patient safety. Pen injectors require patients to manually attach a pen needle, unlike autoinjectors that come with a needle attached. This handheld device enables patients to administer m...
-
Product Pinch clamps
Qosina offers wide range of clamps & clips which includes pinch clamps. Contact us for more information.
-
Product Medical devices & Diagnostic Consummables
Union Plastic proposes custom made medical device. Mold integration and contract manufacturing.
Contact us for more information.
-
Product Disposable Insulin Syringe
Available in 0.3cc/ml, 0.5cc/ml, 1cc/ml, Super sharp 31G, 30G, 29G, 28G Needles, Super low dead space, 10Pcs/bag or 1Pc/pouch Packaging, Sterilized by EO Gas, Comes with two orange color protective caps, CE and US FDA 510K approved, Conformed with ISO5837, Easy and comfortable to use, Latex free gasket.
-
Product IP Creation
For every project Duoject works on, we strive to create a strong IP background to ensure our client’s commercial success for many years to come.

-
Product Hemorrhoid - Arnica - Burn Creams Medical dev. Health & Care
NEW Dermatological medical device creams indicated for the treatment of HEMORRHOIDS relieves irritation, swelling, itching and painfARNICA for muscle pains, oedema, bruises and stings
BURN treats effectively exfoliation, swelling and itching due to burns as well as irritations.

-
Product Medical Device Testing
R&D Analytical SupportIntertek’s significant expertise in bioanalysis and clinical pharmacokinetic studies allows us to support medical device companies working in the areas of preclinical and clinical drug development. Our support services include assay development for API’s, impurities, trace metals ...
-
Product Demopia Demodex - sterile, preservatives free wet wipes with marjoram oi...
What are Demopia Demodex® wipes?
Demopia Demodex® are sterile wipes for professional hygiene of the eyelids, eyelashes and face.
What are Demopia Demodex® wipes used for?
Demopia Demodex® wipes are recommended for the daily care and cleansing of sensitive eyelids, with part...
-
Product Allpresan® diabetic INTENSIVE
Allpresan® diabetic INTENSIVE for very dry to cracked skin
USP´s:
• Medical Device with 10 % Urea, Panthenol and Saccharide Isomerate • Encourages the skin’s healing process and assists with restoration of the damaged skin barrier • Forms a breathab...
-
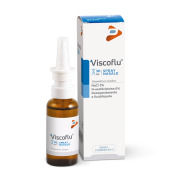
Product VISCOFLU ® nasal Spray
VISCOFLU nasal spray is a medical device made from 3% hypertonic saline solution at controlled pH and 6% N-acetylcysteine. VISCOFLU nasal spray is indicated to facilitate the fluidification and mechanical removal of mucus and/or muco-purulent secretions stagnant inside the nasal cavity (acute...
-
Product GanleePen™(reusable insulin pen)
GanleePen is a reusable insulin pen produced by Gan & Lee's wholly-owned subsidiary, Gangan Medical Technology Co., Ltd. It has user-friendly design, big dose window, precise dose and clear reminder for dose adjustment. In addition, GanleePen is made of high-quality and reusable materials. The color of...
-
Product Molly® Connected Cap Autoinjector
The Molly® Connected Cap is a compact, retrofittable autoinjector add-on that records and transmits data on patients’ use of the device. Upon cap removal from the Molly device, the Connected Cap becomes active, allowing timestamped data to be relayed either through a smartphone app or smart data transmissi...
-
Product Betadex Sulfobutyl Ether Sodium Binzhou Zhiyuan
Product Description
Sulfobutyl ether beta cyclodextrin sodium is a new type of anionic high soluble cyclodextrin derivatives. It could include drug molecules to from covalent compounds so that increase the drug's stability,solubility,safety.It could reduce the renal toxicity,moderate drug's hemolysis,...
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance