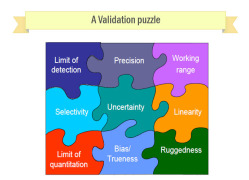
Stabiity Storage and Analysis

Product Description
Oasis Test House

-
IN
-
2018On CPHI since
Categories
Oasis Test House

-
IN
-
2018On CPHI since
More Products from Oasis Test House (2)
-
Product Method Development and Validation services
Method development and validation can be a complex story, and Oasis Test House has systematic procedures to guide its Customers through the process. Our team team of dedicated and qualified personnel are competent to develop methods and validate them as per ICH Guidelines. Before we begin a ... -
Product Microbiological Analysis
Oasis Test House caters to the microbiological requirements for its clients serving them with the below analysis. • Preservative Efficacy Analysis • Sterility Test • Sterility Validation • Microbial Limit Test Validation • Microbial count and pathogens • Bioburden testing • Antibiotics assay • Vitam...
Oasis Test House resources (1)
-
Brochure Oasis Test House Brochure
Find more about our services.Oasis Test House - Your partner in Analytical services Techno-Capabilities Services · Batch Release and 2nd Testing · Analytical Method Development & Validation · Stability Storage and Analysis · Dissolution profiling studies Chemical & Instrumentation Analysis Amino Acid analysis Sugar analysis (Sucrose, Lactose, Mannitol, Sorbitol) In-Vitro Dissolution / Drug Release Studies for conventional and Modified Release Formulations Residual Solvents / Organic Volatile Impurities Particulate matter Elemental Analysis by AAS Microbiological Analysis · Preservative Efficacy Test · Antibiotics Assay · Vitamins Assay · Microbial count & Pathogens · Sterility Test · Bacterial Endotoxins Test (LAL test)
Recommended Products
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance