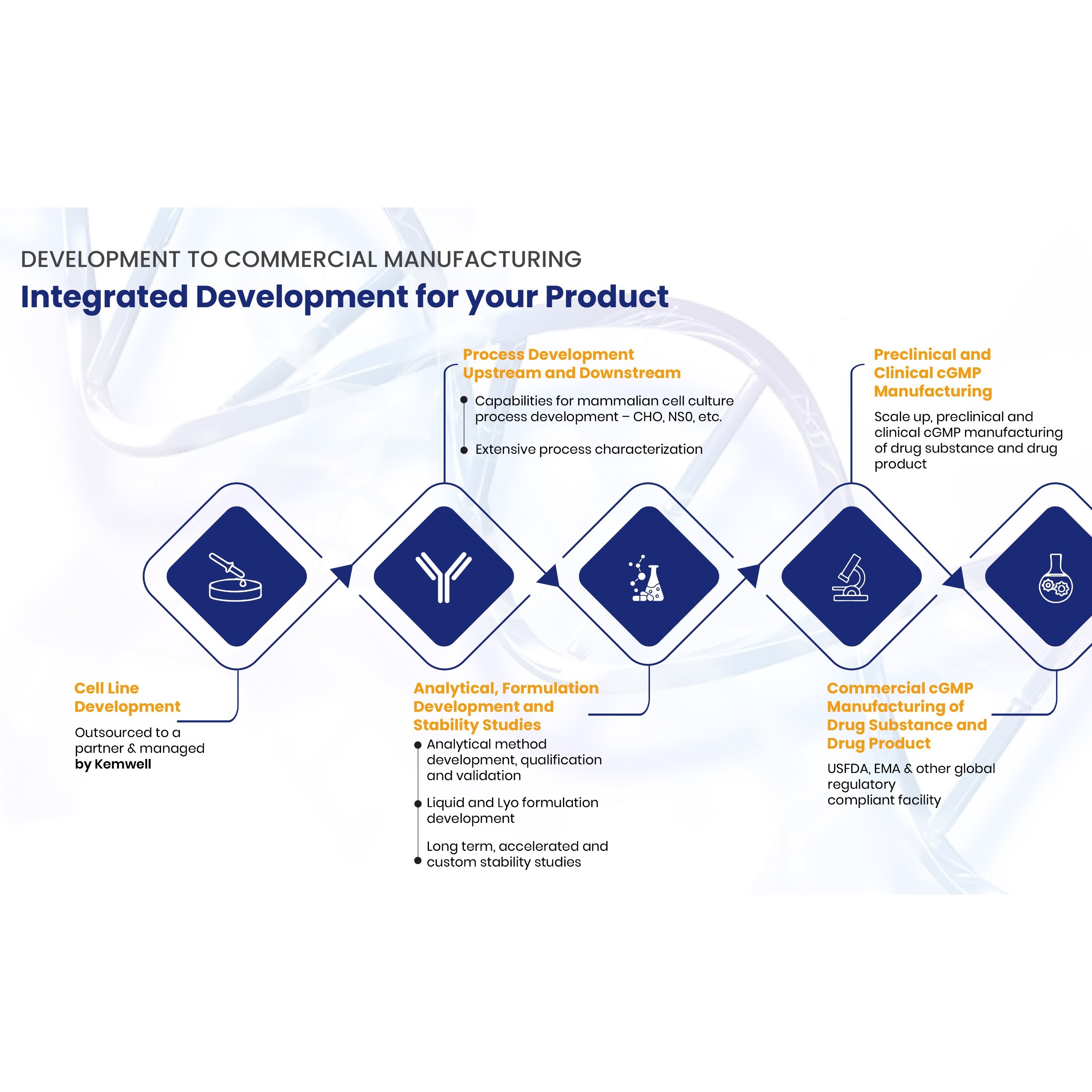
Remsima SC
Product Description
Celltrion
-comp304625.png)
-
KR
-
2022On CPHI since
-
4Certificates
-
1000 - 4999Employees
Company types
Primary activities
Categories
Celltrion
-comp304625.png)
-
KR
-
2022On CPHI since
-
4Certificates
-
1000 - 4999Employees
Company types
Primary activities
More Products from Celltrion (4)
-
Product Remsima
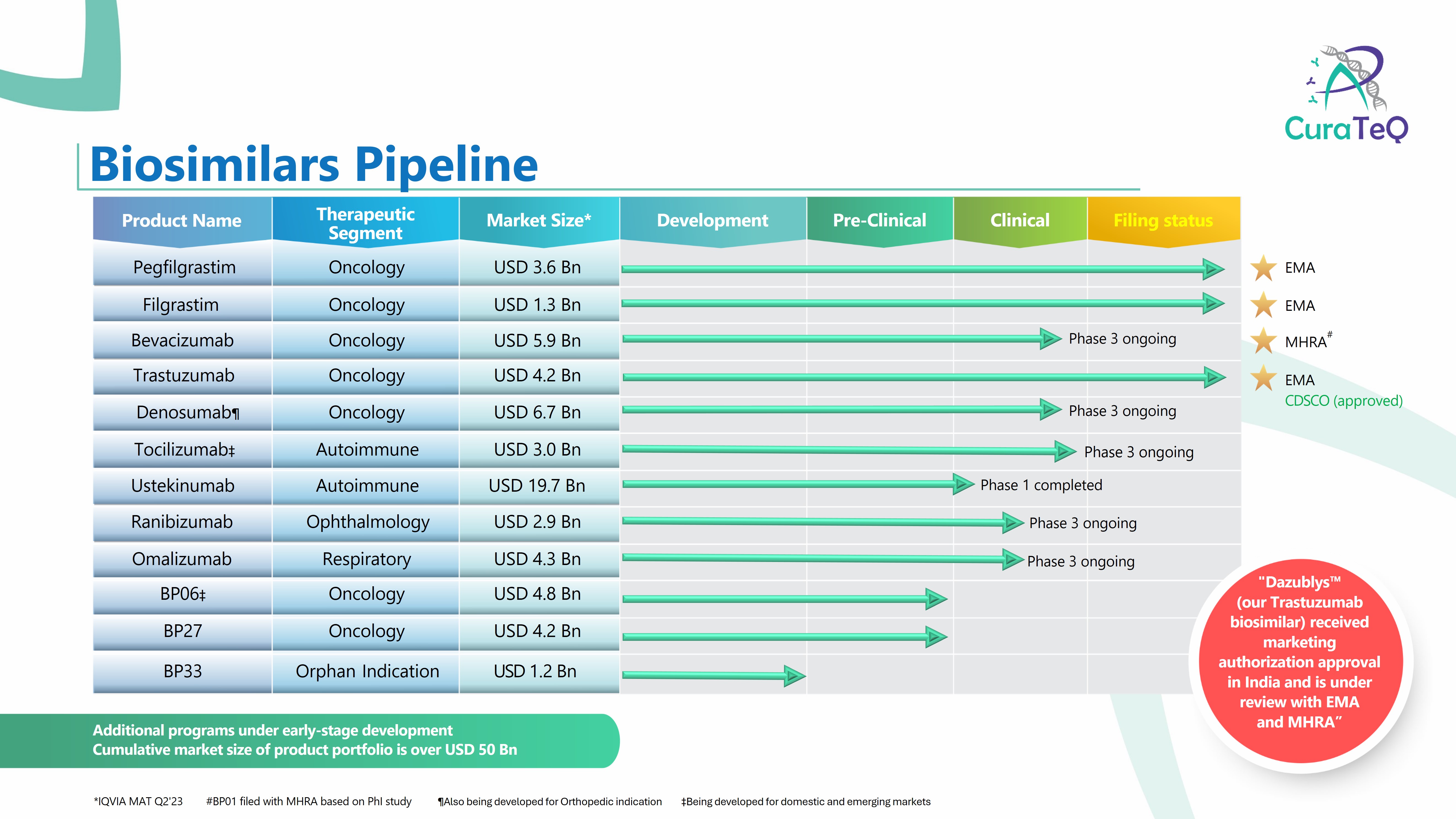
The world's 1st mAb biosimilar • Ingredient: Infliximab • Indication: Rheumatoid arthritis, inflammatory bowel disease • Mode of mechanism: Relieves disease progression by inducing a neutralizing response to tumor necrosis factor(TNF-α), the cause of autoimmune disease • Approval status: 100 countri... -
Product Truxima
• Ingredient: Rituximab • Indication: Non-Hodgkin's lymphoma, rheumatoid arthritis • Mode of mechanism: Specifically binds to CD20, which is expressed on the cell surface of human B cells and lyses target cells to express pharmacological action with mechamisms such as complement-dependent cytotoxicity, ... -
Product Herzuma
• Ingredient: Trastuzumab • Indication: Breast cancer, stomach cancer • Mode of mechanism: Inhibits tumor cell proliferation and growth by binding to a specific antigen (HER2) that occurs specifically and excessively on the surface of tumor cells • Approval status: 80 countries including US, Euro... -
Product Yuflyma
• Ingredient: Adalimumab • Indication: Rheumatoid arthritis, inflammatory bowel disease • Mode of mechanism: Blocks the transmission of inflammatory signals by binding to TNF-α in the body • Approval status: 44 countries including the US(product name: Inflectra), Europe and South Korea
Celltrion resources (3)
-
News Bora Pharmaceuticals partners with Celltrion to crack the APAC market
Bora Pharmaceuticals, the international cGMP CDMO producing specialised pharmaceutical products such as sterile and non-sterile ophthalmics and nasal sprays for clinical use, is partnering with Celltrion Asia Pacific, subsidiary of Celltrion Group Inc. -
News The Korean biopharma market: Incentivizing innovation
With new drug approvals hitting a record high in 2021 and unprecedented number of out-licensing deals closed in 2020, Korean pharmaceutical companies and CDMO’s are poised for significant market growth [i].
Frequently Viewed Together
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance
.png)

























































-comp316899.png)