CPHI Online is the largest global marketplace in the pharma ingredients industry
-
Products0
-
Companies0
-
Articles0
-
Events0
-
Webinars0
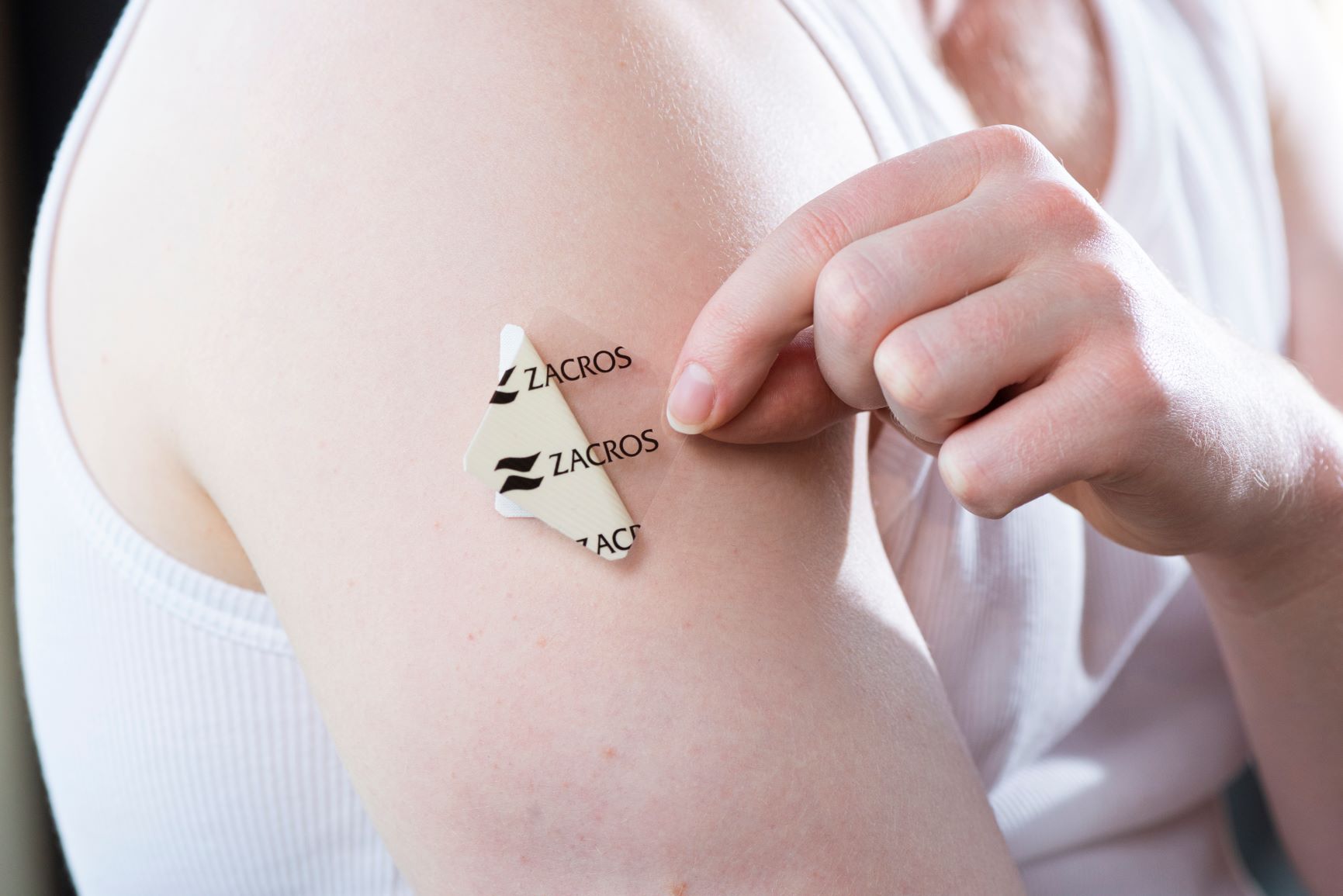
Release liner for transdermal drugs FILMBYNA®
Product Description
FILMBYNA® is a release liner made with
a
PET base film, which preserves the drug agent while offering excellent release
properties for transdermal drug delivery systems.
Designed to provide optimal peel
strength and extended
shelf-life for
a wide variety of adhesives, it protects the surface with stable long-term
performance.
ZACROS has been a leader in release
liners for transdermal drug delivery applications for over 30 years, both in
Japan and globally.
ZACROS has the R&D capacity to
develop custom products to meet specific requirements, and can also provide
primary packaging for patches.
- Long-term stable peel strength
and residual adhesion (6-12 months depending on product)
- Uniform thin-film coating
technology allows for full control of release force design of various adhesives
- Many customization options: printing, coating on one side or both sides (including differing peel
strengths)
- Manufactured in Class 1,000 (ISO
6) to Class 10,000 (ISO 7) clean environment
- DMF and NMPA-registered products
available
ZACROS

-
JP
-
2020On CPHI since
-
3Certificates
-
1000 - 4999Employees
-
-
-
Bunkyo Garden Gate Tower 22F, 1 Chome-1-1 Koishikawa, Bunkyo City, 1120002, Tokyo, Japan
Categories
Specifications
- Supplied fromJapan
ZACROS

-
JP
-
2020On CPHI since
-
3Certificates
-
1000 - 4999Employees
-
-
-
Bunkyo Garden Gate Tower 22F, 1 Chome-1-1 Koishikawa, Bunkyo City, 1120002, Tokyo, Japan
More Products from ZACROS (2)
-
Product MediTect™ IV Bags
Designed to prevent extractables and leachables (E&L) from packaging material, these soft bags are the ideal option for more sensitive drug solutions.
The most important thing to consider in primary packaging is "interaction factor." By using Cyclic Olefin Polymer, an extremely inert materi... -
Product Packaging development service
From design to proof of concept, to small-scale manufacturing, to mass production, ZACROS has extensive experience in co-developing dedicated packaging for pharmaceutical applications.
Through our experience, rest assured that we will develop a highly functional, scalable product for your application.
Recommended Products
-
Product ADAPTEK® TECHNOLOGY
ADAPTEK® TECHNOLOGY is the general name for our 4 international patents and expertise in nanotechnology and controlled release systems.This is a transversal technology that allows to develop customized biopolymeric nanohydrogels, allowing the load with different API (drugs, vitamins, growth factors, etc.)....
-
Product Walk in Cooling Cabinet.
The Walk-In Cooling Cabinets / cold rooms are designed according to ICH guidelines, WHO, MCA and USFDA requirements to maintain uniform conditions. They are superior in airflow distribution, temperature control technology, cabinet construction and are manufactured as per cGMP regulations.
Biggest Walk...
-
Product Omag Packaging Machine mod. CS Junior
CS Junior packaging machine: different products and stick format sizes in one machine. Thanks to its compact design, this machine is the ideal solution for limited space area, and to pack small batches or to test a new market. In only one square meter you can have up to 3.600 stick/hour on one packaging l...
-
Product Pharma Grade Packo jars
Pharma Grade Packo is a screw top jar that has been tested according to both European and US Pharmacopeia standards and is suitable as primary packaging for pharmaceuticals.
Plastic High Performance Packaging by CurTec is clean, safe, and secure and offers maximum protection to active ingred...
-
Product Screw bottle 10ml (Fr4-10)
Screw bottle Fr4-10 with neck 18/415.
Nominal capacity is 10ml.
Bottle is designed to be complemented by a nasal, throat, or fine mist sprayers with a Screw-on cap.
Also available complementation with screw-on cap with PE liner.Pharma grade materials.
Clean room ISO7.
Flexible mi...
-
Product CAPS FOR SINGLE DOSAGE ORAL LIQUID
Capsulit S.P.A offers wide range of products which includes caps for single dosage oral liquids. It is available for two applications one for ready to use medicines and the other for solutions that have to be mixed just before use. It is available in facila ,top facila, facila plast. Contact us for more in...
-
Product Aseptic Blow-Fill-Seal packaging machine for the production of bottles -...
IV solutions are basic life-saving drugs that are as wide ranging as their applications, including hydration agents, chemotherapy, pain management, antibiotics, plasma volume expanders, total parenteral nutrition, and many more. The blow-fill-seal process offers the highest level of sterility assur...
-
Product Advanced PolyPropylene and PolyEthylene Multilayer Infusion Bag films
PolyCine Standard
Multilayer Film for infusion bag production
Packaging for Antibiotics, Anti-Viral Drugs, Haemodialysis Solutions, CAPD Solutions, Standard Infusion Solutions, Plasma-Volume Expander, Anti-Infective Drugs, Analgesics etc.
Customized widths, roll lenghts etc.
qq...
-
Product Pumps for oral and topical applications
RxPack manufactures a wide range of oral and topical pumps with different dosages, closure types and pharma materials in a clean room environment. Different actuator options exist for oral and topical applications.
-
Product ANTI-CELLULITE AND FAT REDUCER PATCH
The product is a patch that has a deep anti-cellulite action.The main objective achieved is to fight the fat accumulated in the deeper layers of skin treated with the product. Its active ingredients act directly on the metabolism of lipids (fucus), helping eliminate excess water retention (caffeine) and ha...
-
Product See-through pouches White Line
Features - innovative bottom web 90 g/m2 polyolefin based material combines properties of nonwovens and polymer films - meets the requirements of DIN EN ISO 11607 and EN 868 - sterile barriere properties testet according to ASTM F1608 and F2638 - printing including indicators according to customer requir...
-
Product Formulation Development
Recipharm offer formulation development services for all dosage forms. We develop everything from simple formulations for early studies to more complex formulations suited for commercialisation.
-
Product Vials made of PP
MEDICOPACK A/S provides a wide range of plastic vials which is including vials made of PP polymer. The material is Medical Grade and fulfills the Ph.Eur. and FDA regulations. PP vials are available in following sizes: 20 ml, 50 ml, 100 ml, 250 ml, 500 ml and 1000 ml. The vials are available in natural and ...
-
Product Ready to Use IV Bags by BAUSCH Consumables
At Bausch Consumables, a division of Bausch Advanced Technologies, we manufacture empty IV bags to meet your specific needs. With our state-of-the-art ISO 7 cleanroom operations and dedicated engineering design team, we deliver high-quality products and exceptional service.
Prompt delivery With stream...
-
Product SELECT
SELECT line:- Metering pumps with doses from 0,25 to 3 ml
- Bottles: from 30 to 250 ml
- Pouches: from 30 to 250 ml.
Contact us for more information.
-
Product Liposomal Vitamin C Beadlets
Liposomal Vitamin C Beadlets
Dosage: 500 MG Vitamin C
Liposomal Vitamin C contributes to the normal functioning of the immune system, the nervous system and to the reduction of tiredness and fatigue. Beside these there are more EFSA claims about vitamin C.

-
Product BFS One-Stop Partner
The Rommelag Group sees itself as a One-Stop Partner for Blow-Fill-Seal Technology and a specialist for flexible Containment Solutions. With our complete solutions for Fill&Finish, we are a strong partner for the pharmaceutical, food, cosmetics and chemical industries.
-
Product Sympfiny®: Delivery device for multiparticulate drugs
Sympfiny® is an innovative drug delivery device for dosing and dispensing multiparticualte drug formulations.Multiparticulates are an emerging oral dosage form particularly suited for use in paediatric medicine. The Sympfiny® system, comprising oral syringe and container, enables caregivers to store, extra...
-
Product Itami patch, Diclofenac sodium salt 140 mg
Fidia Farmaceutici Spa offers a wide range of joint healthcare products which includes itami. Active ingredient(s): diclofenac sodium salt. Indications: local treatment of painful and inflammatory pathologies of rheumatic or traumatic origin of the joints, muscles, tendons and ligaments. Presentation: self...
-
Product DWK Life Sciences Primary Packaging Solutions
DWK Life Sciences is a leading global manufacturer and supplier of glass and plastic primary packaging. Our Primary Packaging container closure systems are manufactured from the highest quality pharmaceutical-grade tubing designed specifically for pharmaceutical, biotech, diagnostic/IVD, and personal ...
-
Product Blister serialization
Although primary pack serialization is not yet a mandatory requirement, it is important to consider the benefits of adopting this technology in view of current healthcare industry production scenarios. Firstly, today the technological evolution of the digital printing industry and the development of new ha...
-
Product BD Vystra™ Disposable Pen
Confidence in dose delivery - BD Vystra™ Disposable Pen is an intuitive, high-quality and customizable pen injector designed to support therapies that require frequent, low-volume injections or variable dosing.
BD partners with you to develop self-injection systems ...
-
Product chemical bottles
Amber glass chemical bottles 50ml - 4400ml. Available in round and square shape with different neck finishes from nerrow neck 32ml to wide neck 80mm.
-
Product Nicotine Transdermal Patch
Nicotine Patch helps you to quit smoking using the principle of Nicotine Replacement Therapy. The patch when placed on your body gradually provides a measured dose of harmless nicotine through the skin over the period of 24 hours. As the nicotine doses are lowered over a course of 12 weeks, the tobacco use...
-
Product Products for film coating, sugar coating, tableting, coloring and excipi...
Excellent excipient premixes for film coating, sugar-coating, coloring and tableting. Tailormade & ready-to-use.
-
Product Measuring Cups and Measuring Spoons
Measuring Cups
Intended purpose
Measuring cups & spoons are used for dosing liquid and/or powdery pharmaceuticals. Our dosing aids can be used to dose and apply pharmaceuticals.
The ml graduation of these dosing aids allows different volumes to be measured...
-
Product OR2Pack®
OR2pack® is an innovative concept for the packaging of sterile implants. Maintaining sterility and traceability of implants during surgery is an absolute necessity to preserve the safety and health of the patient. OR2Pack is a patented solution from ARaymondlife using 2 air-tight square tubes thus allowing...
-
Product Single-dose packaging
TekniPlex Healthcare is specialised in the production of strips of single-dose. The strips can size from 0.3 ml up to 10 ml of product and made with different types of plastic, coloured and customised with embossing, pad printing and silk printing.The system features pre-made unit dose vials is a blow fill...
-
Product Oral Drug Delivery - May/Jun 2023 - Issue 148
Available now to read online at: https://www.ondrugdelivery.com Contents: Strategies for Streamlining the Development of Complex Oral Drug Products Aruna Railkar, Senior Drug Development Consultant. Quotient Sciences Modified-Release Microspheres: Maximising Coating Integrity and Optimising Release Jona...
-
Product Laminated Tubes
Laminate tubes are suitable for cosmetics, pharmaceuticals and food products. They are manufactured from multi-layer laminate foil with the use of Aluminum or plastic barrier that is inserted between the inner and outer layers of the tube. Laminate tubes represent and easy, hygienic and safe method of ...
-
Product Duran Pure Bottles by DWK
Production laboratory, pilot plant, and cell culture production.Biotech, diagnostic, pharmaceutical, CRO, and CMO applications.
Designed for manufacturing of pharmaceutical products, storage of bulk drug intermediates and ingredients, as well as transportation of APIs.
20-20,000 mL sizes available in...
-
Product Rigid Films
Our thermoformable rigid films are ideal for many applications. Think about trays or blisters for surgical kits and medical devices. The films can be equipped with a variety of functionalities and barriers to properly secure its content, ensuring maximum product protection and patient safety.
-
Product IRIS Single-Use Assemblies
Get your customized IRIS single-use assembly & tubing system based on different tubing modules and aseptic connectors directly from Single Use Support. As an expert in single-use solutions, we have made it our goal to deliver vendor agnostic single-use assemblies manufactured with highest qu...
-
Product Complex -Printed for Powder Form
Printed for Powder Form:A commitment to quality and innovation serving the pharmaceutical industry
-
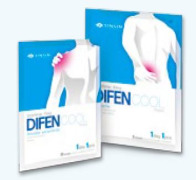
Product Difencool plaster (Diclofenac patch)
Active ingredient : Diclofenac sodiumPacking unit : 10sheets/pack
Characteristics : Pain relieving effect with quick effect after application.
Having adhesiveness with no discomfort in activity while the product is applied.
Usability is improved with easily detachable adhesive sides wh...
-
Product Snowbell Vial Line (Liquid/Powder)
Complete integrated line consisting of : • Vertical rotary vial washing machine
• Sterilizing & depyrogenating tunnel
• Liquid vial filling & bunging machine with statistical / 100% check weighing under isolator
• Transfer conveyors with isolator
• Automat...
-
Product CSTD Ready-to-Use
CSTD (closed system drug transfer device) & CSSTD (closed system sterile drug transfer device).
Drug Reconstitution and intravenous infusion / intravesical instillation systems for various medicines, adhere to the principles of closed circuit, needle-free and contamination-free procedures, elimin... -
Product Cylindrical Bottles
For Special Caps
For Special Spouts and Caps
For DIN 20 Caps
For PFP 28 Caps
Tall Boston Round for 24/410 Caps
For Snap-on Neck D36
For 24/415 Caps
brhrbrhttp://www.neutroplast.com/en/products/cylindrical-bottles/
-
Product EZ-fill® Platform
Many Pharmaceutical companies are looking for ways to lower their footprint and reduce total costs by relying on experienced external partners for non-core activities. When it comes to containment solutions for biopharmaceutical products, no solution matches our EZ-fill®, whose proven advantage...
-
Product Machines For Filling Ampoules And Glass Or Plastic Vials
Rota Verpackungstechnik offers a wide range of products which includes machines tio fill ampoules and glass or plastic vials. Contact us for more information
-
Product Throat spray pump with fixed long nozzle, Meter dosed
Key Features: 1. Neck type: 18GL, 18Din, 18/410, 18/415, 20/410, 24/410 screw on, 20mm snap on, 20mm crimp on.
2. Dosage: 45mcl to 220mcl per stroke, +/-10% tolerance
3. Easy priming and repriming
4. Individual packing available
5. Produced and sealed in Class D clean room
6. ...
-
Product Effervescent Packaging with IML-Technology / Offset Printing or PCR
Sanner is the one-stop-shop supplier for effervescent packaging. No other manufacturer can offer you this range of high-quality system solutions with different decorating techniques such as offset HD or In Mould Labeling : from the smallest Flip Off closures with short tubes through to long vials a...
-
Product Pipette and dosing syringe
Union Plastic offers a wide range of pharmaceutical plastic products which includes pipettes. Contact us for more information.
-
Product Packaging & Serialization
Avéma can help you improve patient compliance with innovative packaging solutions for solid and liquid dose productions. Our facilities offer flexibility to accommodate run size and interchangeable tooling for line flexibility and cost efficiency – so, we can provide support for all products at all stages ...
-
Product Unit Dose Packaging
Tjoapack has been providing unit-dose packaging services to the pharmaceutical industry since 1989. We now supply approximately 85 million unit doses to Benelux hospitals every year. We offer unit dose packaging solutions that allow medicines to verified at the point of administration. By allowing hospita...
-
Product Medical Devices based on substances
We develop and produce medical devices based on substances of class I, IIa and III.
In particular:
Oral Spray
Nasal Spray
Ear Spray
Ear Drops
Oral drops
Oral solids (tablets and sachets)
Vaginal Ovules
Vaginal washes
Eye drops (single an...
-
Product Aluminum printed foil
Aluminium Long experience and top quality to produce primary packaging aluminium for blisters, in two controlled areas.-comp267646.png)
-
Product Aluminium Tubes
For generations, aluminium tubes have been the first-choice packaging for highly sensitive products. Today, more than a century after the tubes were invented, they have lost none of their appeal. No other material protects so effectively against light, air and other environmental factors, and naturally gua...
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance

































.jpg)















.jpg)

![MGDA, Methylglyoxal Dimethyl Acetal [6342-56-9]](https://www.cphi-online.com/46/product/129/72/19/inventys-CPHI_Booth.No_Product-6342-56-9(2024Aug22-v0-CT).png)