Raw Material Services

Product Description
Curida A.S.
-
NO
-
2018On CPHI since
-
2Certificates
-
100 - 249Employees
Company types
Primary activities
Categories
Specifications
Curida A.S.
-
NO
-
2018On CPHI since
-
2Certificates
-
100 - 249Employees
Company types
Primary activities
More Products from Curida A.S. (5)
-
Product Bottle
Curida offers contract development and manufacture of non sterile pharmaceuticals, nutraceuticals and cosmetics in bottles -
Product Blow Fill Seal
Curida offers contract manufacture of liquid pharmaceuticals in Blow Fill Seal ampoules.
Specialized in development activities and full-scale commercial manufacturing of aseptic, sterile and non-sterile fill & finish
Ampoules for small volume units: aseptic filling or terminal ... -
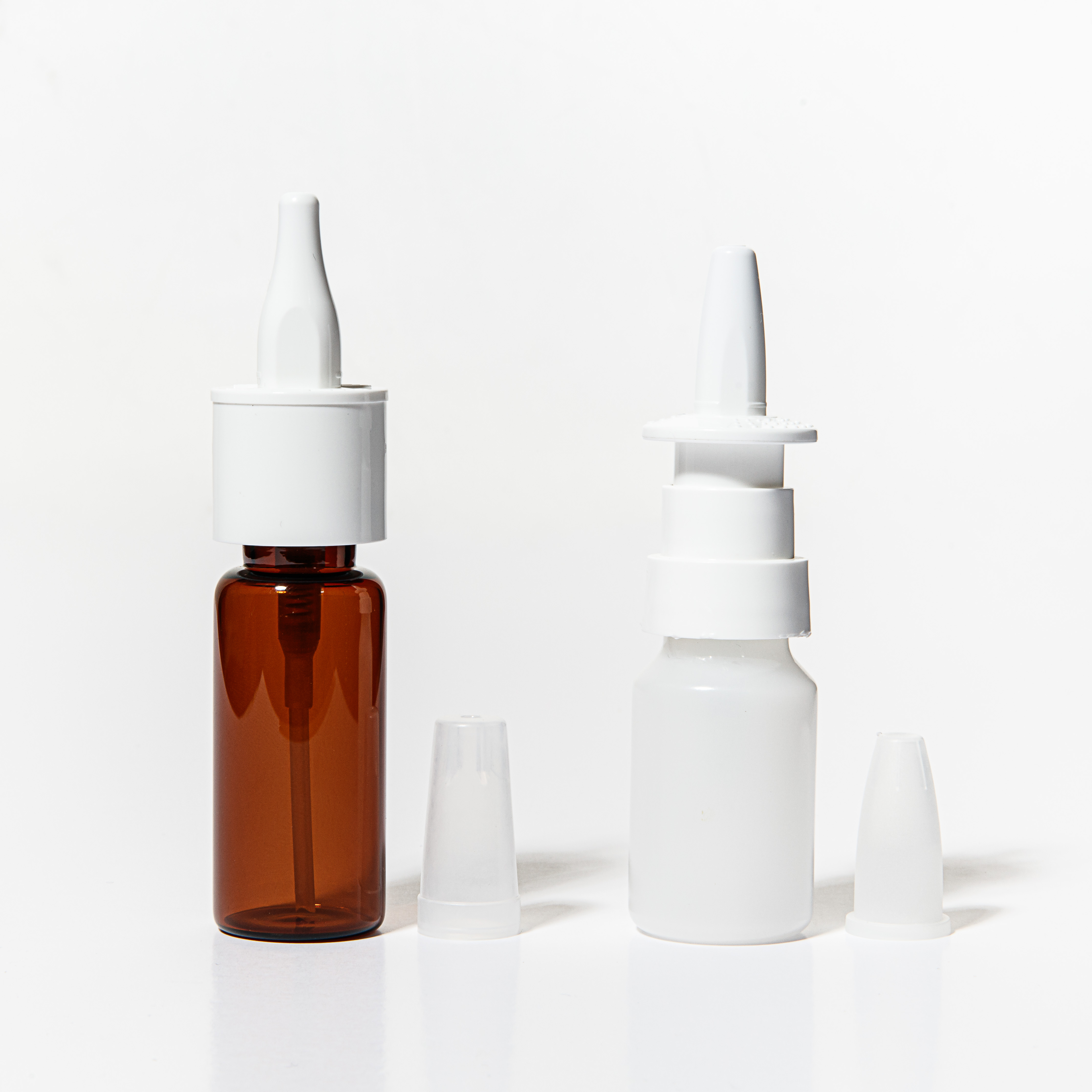
Product Spray
Curida offers Contract Manufacture and Development of non-sterile, single and multi dose sprays.
Cough & cold, allergic conditions, and controlled substances.
Multidose nasal spray for solutions and suspensions -
Product Laboratory Services
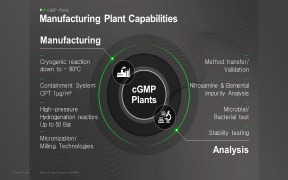
Curida offers Lab services1 chemical lab
3 microbiological labs, including sterility lab and lab for test organisms
Main services:
Quality Control
Method development and validation
Stability testing
Contract laboratory -
Product Monoclonal Antibodies GMP
GMP manufacturing of Monoclonal Antibodies for early-phase through all clinical stages
Also serving the Diagnostic industry with high quality antibodies
Exporting antibodies worldwide, including the US
Frequently Viewed Together
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance















































-comp246698.jpg)