Precious Metal Recovery Service

Product Description
Mastermelt Group Of Companies

-
GB
-
2018On CPHI since
-
1Certificates
-
50 - 99Employees
Company types
Categories
Specifications
Mastermelt Group Of Companies

-
GB
-
2018On CPHI since
-
1Certificates
-
50 - 99Employees
Company types
More Products from Mastermelt Group Of Companies (2)
-
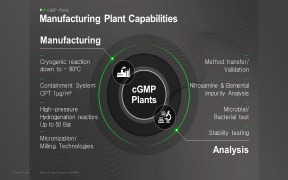
Product Homogeneous Catalyst Precious Metal Recovery
The increased use of Homogeneous catalysts in the pharmaceutical sector for reactions such as hydrogenation, asymmetric hydrogenation, reductive amination, carbonylation, dehydrogenation, isomerisation - Mastermelt have developed Metal Scavenger recovery systems to capture precious metals from these unique... -
Product Precious Metal Recovery from Pharma Reactions
Catalyst characterisation based on an extensive knowledge base and experience, will determine a tailored treatment process, which will deliver the best value returns. Understanding your process where the catalyst is utilised will help determine the treatment route and matrix effects, such as the residual ...
Mastermelt Group Of Companies resources (1)
-
Brochure Circular Economy of Catalyst Processing
Information on the refining services we offer for heterogenous and homogeneous catalyst waste streams
Recommended Products
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance




































-comp246698.jpg)