Merieux NutriSciences | Extractables and Leachables studies
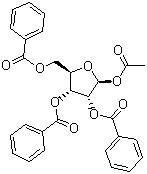
Product Description
Chelab

-
IT
-
2020On CPHI since
-
4Certificates
-
1000 - 4999Employees
Company types
Primary activities
Categories
Specifications
Chelab

-
IT
-
2020On CPHI since
-
4Certificates
-
1000 - 4999Employees
Company types
Primary activities
More Products from Chelab (7)
-
Product Foreign Particles
Merieux NutriSciences has developed various strategies and complementary approaches for the identification of foreign visible and subvisible particles, thanks to sophisticated instrumentation combined with a pool of experts in different fields.Download our brochure: https://bit.ly/48HKnVs -
Product Merieux NutriSciences - Dietary and Food Supplements services
Thanks to specialized laboratories, a GMP pharmaceutical division and a global network of experts at Merieux NutriSciences, we have expanded our expertise in the dietary supplements and food supplements sector. We are able to offer a complete service to support companies in ensuring dietary supplements qua... -
Product Merieux NutriSciences - Medical Device services
Thanks to a valuable pool of experts, Merieux NutriSciences supports you in the development of the testing plan to determine which studies are necessary to ensure that the device is safe and effective, meeting the essential requirements for affixing the CE mark on the product. Download our brochure: ht... -
Product Mérieux NutriSciences | Pharma & HealthCare complete services
Download our brochure: https://bit.ly/4dzryoA
Our complete range of Pharma services include: • Quality controls • Investigation studies • Absorption studies • R&D and validation activities • Stability & storage • Cleaning and disinfectants validation • Environmental monitoring -
Product Merieux NutriSciences | Absorption and efficacy in vitro studies
Download our brochure: https://bit.ly/4dzryoA
In vitro models mimicking different biological barriers can be adopted to perform absorption studies and evaluate efficacy of products, ingredients or new formulations before to proceed with the next step evaluation (e.g. clinical trial).
q... -
Product Merieux NutriSciences | Medical devices services
Download our brochure: https://bit.ly/4dzryoA
Thanks to a valuable pool of experts, Mérieux NutriSciences supports you in the development of the testing plan to determine which studies are necessary to ensure that the device is safe and effective, meeting the essential requirements for affi... -
Product Merieux NutriSciences | Nitrosamines analysis on pharmaceutical products
Download our brochure: https://bit.ly/4dzryoA
Thanks to the long-standing experience, Mérieux NutriSciences has been developing various strategies and approaches for the determination of nitrosamines residues in different matrices through sophisticated mass spectrometry combined with a pool of ex...
Chelab resources (11)
-
News Merieux NutriSciences approach | Nitrosamine alert
Mérieux NutriSciences' approach to addressing the global nitrosamine alert incorporates several innovative elements that significantly advance current solutions on the market, with promising implications for future developments in the pharmaceutical industry. -
Brochure Mérieux NutriSciences | Pharma Service Catalogue
Pharmaceutical Laboratory Services - One-stop shop pharma facility.
Mérieux NutriSciences is a valued partner to the global healthcare industry, offering research, development and quality control testing of pharmaceutical products.
From active ingredients to finished products, we offer a complete range of analytical and research services for API and pharmaceutical products.Our services include safety and quality control, development and validation of analytical methods. Our fully equipped, state-of-the-art laboratories offer comprehensive testing services according to the current pharmacopoeia. We also provide tailored solutions according to customer specifications.
-
News Merieux NutriSciences | Passepartout Studies
We are excited to introduce our Passepartout Studies, a revolutionary approach to extractables profiling for pharmaceutical packaging.
-
Brochure Merieux NutriSciences | Nitrosamines analysis
Thanks to the long-standing experience, Mérieux NutriSciences has been developing various strategies and approaches for the determination of nitrosamines residues in different matrices through sophisticated mass spectrometry combined with a pool of experts.
Our capabilities: The R&D department of Mérieux NutriSciences | Pharma and Healthcare has developed and/or validated high sensitive methods suitable for more than 70 NDRSIs and small nitrosamines. -
Brochure Mérieux NutriSciences | Viral Clearance
Our capabilities - The Viral Clearance Study
Our dedicated virology lab control the viral safety of your product by assessing the capacity of the production processes to clear infectious viruses. The objective of viral clearance studies is to evaluate the ability of the manufacturing process to inactivate/remove known or even unknown viral contaminants, and to estimate process robustness by characterizing its ability to clear different model viruses. -
Brochure Mérieux NutriSciences | Extractables and Leachables studies
Mérieux NutriSciences performs E&L studies providing a full integrated testing strategy together with toxicological assessment and risk analysis.
Extractables and Leachables studies provide a full-integrated testing strategy together with toxicological assessment and risk analysis, in six main steps:Profiling of extractables: generation of the extractCharacterization of extractablesPrimary and secondary leachables profileUnknown extractables/leachables tentative identification by HRMS techniques (if needed)Toxicological evaluation and risk assessmentDevelopment and validation of targeted methods suitable for the quantification of critical leachables -
Brochure Mérieux NutriSciences | Foreign particles identification
The presence of foreign particles in sterile pharmaceutical products and/or medical device can affect their safety and efficacy.Foreign particles may originate from both organic and inorganic sources, as corroded or damaged equipment parts, cross contamination during the process, microplastics or from biological sources. The implementation of quality controls for particles can be a good strategy in order to minimize the contamination probability.
Mérieux NutriSciences has developed various strategies and complementary approaches for the identification of foreign visible and subvisible particles, thanks to sophisticated instrumentations combined with a pool of experts in different fields. -
Video Pushing Boundaries in Nitrosamines Analysis: Spotlight on Nitroso Drug Substance-Related Impurities
The global alert on nitrosamines has profoundly impacted the pharmaceutical industry, necessitating an immediate and comprehensive response from all stakeholders involved. Nitrosamines, which are classified as probable human carcinogens, have historically been associated with the consumption of cured meats and tobacco products, but their unexpected presence in pharmaceuticals prompted a swift reaction from health authorities worldwide. In response to the global nitrosamine alert, Mérieux NutriSciences' Pharmaceutical and R&D Divisions have utilized their extensive expertise to develop advanced methodologies to tackle the challenges posed by nitrosamine impurities. Drawing on a strong background in the chemical industries, their experts have pioneered the use of sophisticated mass spectrometry technologies to improve the detection and quantification of nitrosamines in drug products, aiding pharmaceutical companies in compliance efforts. This year, Sergio Fasan, Business Director will present all the latest research and innovation in our pharmaceutical labs: > NDSRIs emerging issues > Analytical challenges during method development and QC tests on NDSRIs > Case studies from MxNS Pharma Nitrosamines Labs -
Brochure Merieux NutriSciences | Absorption and Efficacy in vitro studies
In vitro models mimic the different biological barriers and target organs by which it is possible to evaluate absorption and efficacy of formulations.
Absorption studies permit the performing of several screening tests to preliminarily evaluate products, ingredients or formulations before proceeding with the next steps evaluation. -
Brochure Merieux NutriSciences | Novel Food: the entire value-chain in our labs
Mérieux NutriSciences supports you from the initial assessment of your project to the correct identification of the requirements for submitting the dossier to the Commission.We perform all the necessary tests in our labs,and apply for Novel Food authorisation. -
Brochure Merieux NutriSciences | Medical Devices: innovative Solution for the Healthcare
Thanks to a valuable pool of experts, Mérieux NutriSciences supports you in the development of the testing plan to determine which studies are necessary to ensure that the device is safe and effective, meeting the essential requirements for affixing the CE mark on the product.Regulatory support and Risk AssesmentQuality Controls and StabiltyBiocompatibilityAbsorption and efficacy studiesHuman factor services
Frequently Viewed Together
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance






































































-comp246698.jpg)








































![6-broMo-2-chloro-8-cyclopentyl-5-Methylpyrido[2,3-d]pyriMidin-7(8H)-one](https://www.cphi-online.com/46/product/130/26/06/hy-32205.gif)