Merck Millipore Launches Lockable ICRplus Settle Plates for Safe and Efficient Viable Air Monitoring and Personnel Monitoring
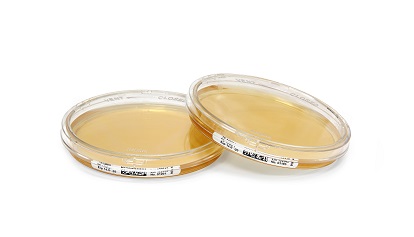
Merck Millipore, the Life Science division of Merck, has launched lockable ICRplus Settle Plates for viable air monitoring and personnel monitoring in clean rooms and isolators.
The new plates are based on the original Merck Millipore ICR Settle Plate technology and provide semi-quantitative determination of microbial contamination. ICRplus Settle Plates feature a new two-way closure system: the 'CLOSED' position allows safe transportation and aerobic incubation while the 'VENT' position expands incubation for the detection of anaerobic and microaerophilic micro-organisms.
ICRplus Settle Plates are filled with Casein Soybean Digest Agar (TSA), which is recommended by FDA Guidance for Industry (2004): Sterile Drug Products Produced by Aseptic Processing — cGMP. The plates are offered in formulations with neutralizers for the inactivation of disinfectant. In addition, the plates can be used to detect fastidious bacteria when used in combination with Chocolate Agar and lecithin, Tween 80 and histidine (LTH).
“Efficient and reliable environmental monitoring of isolators and clean rooms is essential to support product quality and customer safety,” said Yvonne Hochheimer, Product Manager at Merck Millipore. “With our unique two-way closure system of the ICRplus Settle Plates, customers can be assured of safer transport and reliable detection of aerobic and anaerobic micro-organisms. The plates also offer the ultimate in convenience with room temperature storage at the site of use and a long shelf life. This translates into decreased storage costs and fewer incoming goods controls and QC testing.”
ICRplus Settle Plates are produced under aseptic conditions, gamma-irradiated and triple-bagged, making them optimal for personnel monitoring, passive air sampling and active air sampling with sieve-like samplers (for example, MAS air samplers) in isolators and clean rooms. Furthermore, the new products enable inactivation of residual vaporous hydrogen peroxide (VHP) during active air sampling. Each ICRplus Settle Plate contains a data matrix barcode, which allows secure identification of plates and supports the 'paperless' lab. The 90-mm plates have a high filling volume of 30 mL, which allows prolonged incubation.
Related News
-
News Lessons from CPHI Milan 2024: Sunny Intervals for Pharma Manufacturing?
As the 2024 CPHI conference wrapped up in Milan, we caught up with L.E.K. Consulting – a global strategy consulting firm with deep expertise in pharma manufacturing – to discuss evolving market perspectives and business outlook. -
News US BIOSECURE Act passed by US House of Representatives
The controversial act, which has already impacted several foreign companies operating in the US, was passed by the House of Representatives on September 9, 2024. It is now headed for the US Senate before it can be signed into law by President Joe Biden... -
News Pharma Supply Chain People Moves
The latest appointments, promotions, and structural changes across the pharmaceutical supply chain. -
News Drug prices agreed upon as part of the US Inflation Reduction Act
The Inflation Reduction Act brought into constitution by the Biden administation in 2022, which proposed a drug price negotiation between the government and pharmaceutical companies, has reached it's first agreement. -
News BIOSECURE Act continues to loom over Chinese pharma manufacturers
With the US BIOSECURE Act on its way to passing into legislation, Chinese companies are facing declining revenues within the first half of 2024 as US pharmaceutical and healthcare companies pull their businesses from the country. -
News Ophthalmologic drug product Eylea faces biosimilar threats after FDA approvals
Regeneron Pharmaceutical’s blockbuster ophthalmology drug Eylea is facing biosimilar competition as the US FDA approves Biocon’s Yesafili and Samsung Bioepis/Biogen’s Opuviz. -
News ONO Pharmaceutical expands oncology portfolio with acquisition of Deciphera
ONO Pharmaceutical, out of Japan, is in the process of acquiring cancer-therapy maker Deciphera Pharmaceuticals for US$2.4 billion. -
News First offers for pharma from Medicare drug price negotiations
Ten high-cost drugs from various pharma manufacturers are in pricing negotiations in a first-ever for the US Medicare program. President Biden’s administration stated they have responded to the first round of offers.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance




.png)
.png)

.png)