CPHI Online is the largest global marketplace in the pharma ingredients industry
-
Products0
-
Companies0
-
Articles0
-
Events0
-
Webinars0
GCP Services
Product Description
We offer a range of GCP services, from monitoring to audits. Providing comprehensive GCP compliance services for the pharmaceutical industry is one of our specialties.
A. Risk-Based Monitoring:
✔ Risk-Based Monitoring for Bioequivalence/Bioavailability Studies (BE/BA Studies)
✔ Risk-Based Monitoring for Phase-I Studies.
✔ Risk-Based Monitoring for Patient-Based PK/PD Studies.
✔ Risk-Based Monitoring for Late Phase Clinical Trials (Phase-II, Phase-III, Phase-IV)
✔ Expert Monitoring for Bioanalysis (including In-Process and Retrospective)
✔ Expert Monitoring for PK-Stat (Pharmacokinetic Statistics)
B. Audits:
✔ CRO Qualification/Requalification Audits
✔ Pre-Inspection readiness Audit/Mock-Inspection
✔ For-Cause Investigative Audit
✔ Retrospective Study Data Audit
✔ QA Audit
✔ Investigator Site Audit
✔ Safety Assessment Audit
C. Sponsor Oversight: Complete Management of BE/BA Studies/Trial Outsourcing.
✔ Pre-Study Support
✔ During-Study Support
✔ Post-Study Support
D. Standalone FSP Services:
✔ Medical Writing Services
✔ Regulatory Approval Support
✔ CRC/CRA Support
✔ Data Management Services
✔ GCP/GLP Training
✔ Pre-Clinical Facilitation
Zenovel

-
IN
-
2021On CPHI since
-
1Certificates
-
25 - 49Employees
-
-
-
Zenovel, 1113 Satyamev Eminence,, Science City Road, 380060, Ahmedabad, Gujarat, India
Specifications
- Selling PointsBrand Name; Convenience; Experienced Technicians; Guarantee/Warranty; Quality Service; Reputation
- ModelClinical Research/Trials
- Supplied fromIndia
Sales Markets
- Middle East Region (e.g. UAE)
- Oceania
- North America (USA, Canada)
- Central America (e.g. Mexico)
- East Asia (e.g. China, Japan, Korea)
- Europe - EU countries
- Europe - non EU (e.g. UK, Russia, ex-CIS countries)
- South America (e.g. Brazil, Colombia)
- South Asia (e.g. India, Pakistan, Sri Lanka)
- South East Asia (e.g. Thailand, Philippines, Singapore)
Zenovel

-
IN
-
2021On CPHI since
-
1Certificates
-
25 - 49Employees
-
-
-
Zenovel, 1113 Satyamev Eminence,, Science City Road, 380060, Ahmedabad, Gujarat, India
More Products from Zenovel (2)
-
Product GMP Audit & Consulting Services
We aim to provide the most reliable and preferred service platform to pharmaceutical companies worldwide for R&D cycle, regulatory affairs, and GMP compliance with a higher benchmark of quality, integrity, and ethics.
... -
Product Regulatory Support
Zenovel has a highly experienced team of experts who have a complete understanding of the complete requirements of Pharma Regulatory affairs throughout the drug life cycle. Our experts have experience in directly corresponding with Pharma regulatory authorities for various segments.
...
Recommended Products
-
Product Clinical Trial Services
Our end-to-end global clinical supply chain services, formerly Fisher Clinical Services, are powered by people with an unwavering dedication to serving clinical research and patients around the world. Whether your investigational medicinal product (IMP) is a traditional small molecule, biologic, or advance...
-
Product Medical Device Testing
1) Physico-Chemical Testing of Raw Material & Finished Medical Devices2) Biocompatibility Testing of Medical Devices(As per ISO 10993-1:2018)
3) Biological Testing of Raw Material of Plastic, Rubber,Silicone, Polymers, etc.(As per IP / BP / EP / JP / USP)
4) Microbiological Testing Services ...
-
Product Custom Protein Synthesis
We provide a Custom Protein Synthesis Service, using a chemical method that synthesises proteins amino acid by amino acid and making modifications on an atomic-scale. We work closely with our partners in designing, customising and optimising the proteins that is synthesized in an automa...
-
Product Cell-based Neutralization Assays
Experienced cell-based neutralizing antibody assay capabilities. Our experts, at our GLP/GCP compliant laboratory, evaluate each therapeutic, conduct tailored assay development and ensure that each assay is optimized to suitably determine the presence of NAb with maximum possible level of drug toleran...
-
Product Drug efficacy, In vitro and in vivo studies
We offer a full range of studies and bioanalytical services, providing powerful tools and rigorous solutions for preclinical drug development of therapeutic candidates, advance lead compounds towards first-in- human trials, and support for ongoing clinical development
-
Product Data Sciences
You need data rapidly to make crucial decisions during a study. Faster data means we can provide our customers with earlier interim data and interim PK reports for on-study dosing and formulation decisions.
We understand on-study changes to early phase study designs and d...
-
Product Clinical solutions: Trials
Conscio's in-house clinical Unit has been operating since 2007, with more than 600 BA/BE/PK studies completed and proprietary of over 270 validated bioanalytical methods. Equipped with a 60-bed on-site clinical unit in Prague and a recently opened 36-bed unit in Ostrava, we provide comprehensive services w...
-
Product Clinical Manufacturing
KBI Biopharma offers a broad range of cGMP biologics manufacturing services to biopharmaceutical companies worldwide. Our capabilities include reliable manufacturing for preclinical and clinical supply.
KBI’s experienced team produces high-quality therapeutics and vaccines thro...
-
Product Cortellis Regulatory Intelligence™
Navigate the regulatory landscape and efficiently drive strategic decisions with Cortellis Regulatory Intelligence. Get full and timely coverage of global health authority requirements on healthcare products such as drugs, biologics, medical devices and IVDs, and access exclusive expert reports for both in...
-
Product tiny-TIMsg
The tiny-TIMsg system, as part of the SurroGUT™ platform, precisely mimics the upper GI tract and all physiologically relevant processes; including the pressure, shear forces and phase specific gastric emptying. The tiny-TIMsg consists of an advanced gastric compartment (TIMagc) and small intestine co...
-
Product PharmaLex
PharmaLex is one of the largest providers of Development Consulting, Regulatory Affairs, Quality Management & Compliance and Pharmacovigilance worldwide. Our global teams of experts can take you through early strategic planning activities and nonclinical requirements to clinical deve...
-
Product R&D Services
Our R&D Services are categorized into 8 streams: • Material Science Services
• Metal and Organic Scavenging Screening Services
• Organic Synthesis Services
• Catalysis Services
• Process Services
• Chromatography and Purification Services
• Method D...
-
Product Clinical Research Solutions
We design and execute clinical trials so new therapies get to market in the quickest and safest way possible. Full-Service Solution: We manage and execute full-service clinical trials across major therapeutic areas within medical device, diagnostics, drug, and biologic. Functional Service Provisi...
-
Product Integrated Services
Huayi Pharmaceuticals offers an integrated service for product development with our partners Weida Clinical Research and Velocyte Analytical Technology for your bioequivalence needs in China. Through these close strategic partnerships, we are able to offer a one-stop service which allows pharmaceutical dev...
-
Product Drug Product Development
We support our clients from early-stage research and preclinical phase to and beyond market approval by providing comprehensive development services, including
• Developability assessment • Pre-formulation screening • Formulation development • Lyophilization process development • Forced d...
-
Product Microbiology
• Sterility Testing • Quality Control Release • Bioburden Testing • Method Suitability • Microbial Identification - MALDI-TOF • Bacterial Endotoxin Testing • Cytotoxicity • Environmental Monitoring • Biological Indicator Enumeration
-
Product Drug Product Manufacturing Service
Ardena offers services under brand pharmavize which includes drug product manufacturing service. It can manufacture various dosage forms, including: solutions and (nano-)suspensions, powder and granule filled capsules, semi-solid and, liquid filled capsules, topical products, tablets, modified-release prod...
-
Product R & D Laser
A small and powerful manually operated tablet laser drilling and writing machine for products of all shapes and sizes. Accommodates tablets, capsules or soft gels and manages multiple recipes. From laser marking soft gels to laser drilling tablets for controlled release, it delivers consistent, high qualit...
-
Product Formulation Development
Recipharm offer formulation development services for all dosage forms. We develop everything from simple formulations for early studies to more complex formulations suited for commercialisation.
-
Product Certified toxicological studies
Toxicological tests performed at the Lukasiewicz - IPO:
-allow a toxicological evaluation of chemical substances contained in pharmaceutical products, veterinary medicinal products, plant protection products, food and feed additives, industrial chemical substances, etc.,
-make it possi...
-
Product Analytical Services
With 55+ years of experience, our expert teams develop 1,000+ analytical methods and validate 250+ methods annually. Drawing upon an extensive range of analytical technology, combined with a wealth of analytical knowledge, we can add real value to your drug development and commercialisation programs....
-
Product Pre-Clinical Services
Our 3 platforms available are: • Solubility • Solid Dispersion • Nanoparticles
-
Product Clinical Trial Design
Study design is a significant phase of any clinical research project. At Atlantia we feel that our careful planning and guidance to our clients coupled with our extensive experience with regulation, pre diseased and diseased populations, is what brings our clients back time and again to trust us with their...
-
Product Preclinical Functional Neuroimaging
neuroPhINDr is our answer to the problem of low translational value of preclinical neuroimaging. With neuroPhINDr, we aim to shorten time-to-market, increase confidence in preclinical results and improve success rates in phase I trials. neuroPhINDr is a set of highly optimised and standardised protocols fo...
-
Product GCP Services(Good Clinical Practices)
We offer various GCP services which can help the clients across globe, ranging from audits to monitoring services. We provide comprehensive GCP Quality Compliance services to the pharmaceutical industry. Our GCP consulting services are characterized by:
1) Knowledge: Various regulatory requi...
-
Product BDD Swift - adaptive clinical studies
•SWIFT accelerates evaluation and optimisation of product performance using in vivo clinical data to direct formulation design in real time.
•SWIFT studies enable you to make decisions on formulation changes based on emerging clinical data. • This integration of formulation development, GMP manufactu...
-
Product Manufacturing Services
At Sharp, we work closely with you to build an in-depth understanding of your goals, challenges and requirements before we begin our state-of-the-art manufacturing process. This enables us to tailor our clinical manufacturing services to meet your specific needs, making sure we deliver your product on time...
-
Product Clinical Supply Manufacturing
NCEs • Phase I-III formulation process development and clinical supply manufacturing Generic drugs
• BE sample manufacturing • Process Optimization
-
Product Clinical Trial Services
Our experts will accomplish your clinical objectives by leading the way from recruitment strategies, site selection, and clinical logistics, all the way through the regulatory process.
We use our own eClinical suite to be more cost-efficient and keep you connected remotely acros...
-
Product CRO Services
Clinical Development Program (Phase I to IV) Product Registration
Pharmacovigilance
Regulatory Affairs
Clinical Monitoring
Medical Monitoring
Project Management
Medical Writing
Quality Assurance - GCP Audits
Logistics Coordination

-
Product UNITYai.Lit
Elevate your literature management with Unityai.Lit - the advanced software platform and the ultimate solution for efficient and validated ICSR processing and identification of publications for signaling and aggregate reports. Developed by Soterius, Inc., Unityai.Lit is not just a tool; it's a transformati...
-
Product Biomarker Assays and Validation
Validation of biomarker assays: Our diverse range of analytical platforms supports biomarker analyses including ELISA, flow cytometry, cell-based assays, multiplex ECL platforms, and high sensitivity measurements using QuanterixSimoa HD-X Analyzer™ measurement instruments. Our dedicated biomarker assay...
-
Product Orphan Drug Development
Accelerating orphan drug development from candidate selection to commercial manufacture and supply.
With over 300 million people worldwide living with identified rare diseases, the development of new treatments to address these unmet clinical needs is an area of great interest in the pha...
-
Product Cortellis Clinical Trials Intelligence™
Confidently and efficiently plan and design clinical trials by reducing time spent compiling and analyzing protocols, sites, outcomes, endpoints and more. Avoid costly protocol amendments, recruitment delays and trial failure by tracking competitive trials, selecting experienced sites, defining the right e...
-
Product Netpharmalab Consulting Services
Contract Research Laboratory. Analytical Services (physicochemical analysis, microbiological analysis and stability studies) and EU Import Lab.
-
Product Biocompatibility of Medical Devices according to ISO 10993-1 series
Galileo Research can drive the nonclinical development of your medical device by preparing the Literature Report and the Biological Evaluation Plan (BEP) according to ISO 10993-1, performing additional tests, if needed, and releasing the Biological Evaluation Report (BER).
All tests can be condu...
-
Product Outsourcing
Patheon by Thermo Fisher Scientific has a broad manufacturing platform for pharmaceutical and biologic products which provides sustainable solutions for mammalian cell-based and microbial-based manufacturing, green chemistry R&D and manufacturing technologies, and finished dosage production of biopharm...
-
Product Clinical Trial Reporting
The Atlantia Clinical Research team provides a comprehensive study reporting service. Our statisticians adhere to ICH-GCP standards to produce a statistical study report, and if the client requests, a full clinical study report.Atlantia maintains no rights to Intellectual Property generated from this study...
-
Product TIM-2
The TIM-2, as part of the SurroGUT™ platform, is the dynamic in vitro model of the large intestine. The lower GI model, with its anaerobic compartment containing highly metabolic active intestinal microbiota, precisely mimics the physiological conditions of the colon in vivo. TIM-2 can simulate the pr...
-
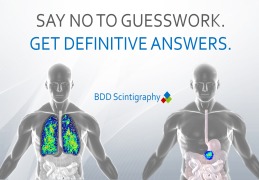
Product BDD Scintigraphy
Gamma scintigraphy is a powerful and versatile technology used to investigate the in-vivo performance of your drug or product. This non-invasive imaging technique allows us to follow the fate of the dosage form and administering it to a volunteer or patient. Not limited to just oral dosage forms, gamma sci...
-
Product 2-8°C Clinical Trials, Biologics, Vaccines
Cold Chain expertise.
CIT can pack any farmaceutical form at 2-8°C
Vials, ampoules, bags,...
Ask for our developed service of 2-8°C monitoring via blockchain

-
Product Bioanaysis
Delivering rapid bioanalytical data in discovery – preclinical – clinical. Bioanalysis is a critical step in the drug development process. At Quotient, we are experts in the development, validation, and application of bioanalytical assays.
Our dedicated team of bioanalytical chemists...
-
Product CRO and CDMO Solutions
CRO and CDMO solutions: Transforming the pharmaceutical value chain for emerging biotechs and large pharmas - How integrating research and manufacturing processes enhances efficiency and accelerates time to market.
-
Product Clinical Supply Solutions Service
Ardena offers services under brand pharmavize which includes clinical supply solutions service. It has a promising drug molecule and it is preparing for preclinical or early phase clinical studie. Its business is to develop and manage the clinical supply materials, from design to delivery. It bring the api...
-
Product GMP Audit & Consulting Services
We aim to provide the most reliable and preferred service platform to pharmaceutical companies worldwide for R&D cycle, regulatory affairs, and GMP compliance with a higher benchmark of quality, integrity, and ethics.
...
-
Product Computer System Validation (CSV) Solutions
ProPharma's validation professionals leverage the latest risk-based Computer System Validation (CSV) and Computer Software Assurance (CSA) techniques to ensure that our clients' systems are ready for inspections from the FDA, EMA, MHRA, and other regulatory agencies. Our consultants have extensive experien...
-
Product Clinical Pharmacology
When you are looking for a partner who is dedicated to Phase I trials and early clinical development, rely on Quotient Sciences for the experience and expertise to seamlessly deliver your studies with the highest quality service and speed. We accelerate your molecule from first-in-human to proof-of-concept...
-
Product Regulatory Support
Zenovel has a highly experienced team of experts who have a complete understanding of the complete requirements of Pharma Regulatory affairs throughout the drug life cycle. Our experts have experience in directly corresponding with Pharma regulatory authorities for various segments.
...
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance





.png)
.jpg)






%20(2).png)





































.png)