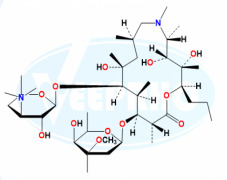
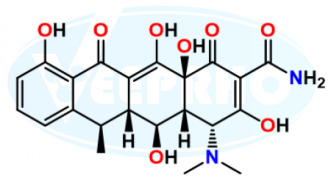
Fulvestrant EP Impurity E

Product Description
Veeprho Pharmaceuticals s.r.o.

-
CZ
-
2019On CPHI since
-
2Certificates
-
100 - 249Employees
Company types
Categories
Specifications
Veeprho Pharmaceuticals s.r.o.

-
CZ
-
2019On CPHI since
-
2Certificates
-
100 - 249Employees
Company types
More Products from Veeprho Pharmaceuticals s.r.o. (2)
-
Product Azithromycin EP Impurity O
Name:Azithromycin EP Impurity OCatalogue No.VL1860010CAS No.763924-54-5Molecular FormulaC39H74N2O12Molecular Weight763.01StatusIn-stockIUPAC Name(2-desethyl-2-propylazithromycin) -
Product Doxycycline EP Impurity C
Doxycycline EP Impurity C / 4-Epidoxycycline / Doxycycline Related Compound C
Catalogue No: VL159001
Molecular Formula: C22H24N2O2
Molecular Weight: 444.43
Status: In-stock
IUPAC Name: (4R,4aR,5S,5aR,6R,12aS)-4-(Di...
Veeprho Pharmaceuticals s.r.o. resources (1)
-
Brochure Company Brochure
Company brochure of our sister company Veeprho Laboratories Pvt. Ltd. where all our products are manufactured.
Recommended Products
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance