- Home
- Rihim Pharma Consultancy Pvt Ltd
- Fixed Dose Combination
CPHI Online is the largest global marketplace in the pharma ingredients industry
-
Products0
-
Companies0
-
Articles0
-
Events0
-
Webinars0
Fixed Dose Combination

Product Description
Rihim Pharma Consultancy Pvt. Ltd. offers a wide range of services which includes fixed dose combination. It is becoming increasingly important from a public health perspective. Such combinations of drugs are being used in the treatment of a wide range of conditions and are particularly useful in the management of hypertension, diabetes, hiv/aids, malaria and tuberculosis, which are considered to be the foremost infectious disease threats in the world today. It offers services such as strategy development for fdc introduction; rationale & justification for fdc; bioequivalence study; etc. Contact us for more information.
Rihim Pharma Consultancy Pvt Ltd

-
IN
-
2015On CPHI since
Company types
Contract Service
Contact info
-
-
518-519, Ring road Mall, Mangalam Place, Rohini Sector 3, Delhi, India
Categories
Specifications
- Supplied fromIndia
Rihim Pharma Consultancy Pvt Ltd

-
IN
-
2015On CPHI since
Company types
Contract Service
Contact info
-
-
518-519, Ring road Mall, Mangalam Place, Rohini Sector 3, Delhi, India
More Products from Rihim Pharma Consultancy Pvt Ltd (2)
-
Product Ba/Be For Export
Rihim Pharma Consultancy Pvt. Ltd. offers a wide range of services which includes ba/be for export. It provide services in ba/be for export from feasibility of getting be-noc to final approval at the office of dcg (i), fda bhawan. It includes services such as updating clients regarding new gazette notifica... -
Product Gct (Global Clinical Trial)
Rihim Pharma Consultancy Pvt. Ltd. offers a wide range of services which includes gct (global clinical trial). It offers such service as the pharmaceutical market experiences rapid growth globally so more clinical trials are being conducted in india and inturn offering new opportunities for pharma industry...
Recommended Products
-
Product Custom Protein Synthesis
We provide a Custom Protein Synthesis Service, using a chemical method that synthesises proteins amino acid by amino acid and making modifications on an atomic-scale. We work closely with our partners in designing, customising and optimising the proteins that is synthesized in an automa...
-
Product e-Reg: management of Regulatory Affairs' big data
OP Pharma has been providing all-around services in the Regulatory Affairs field for more than 20 years to small to big pharma Companies in Italy as well as in the European Union.
Do you have lots of products and/or products' data which you cannot organize/archive interna...
-
Product Drug Development Consulting
Our Drug Development Consultants work with you to design and implement successful drug development programs. With industry leading scientific expertise across a range of technical disciplines, we help customers across all stages of development, from candidate selection to commercial launch.
Our cons...
-
Product QUALIPHARMA
Qualipharma is a consulting company created in 2001 and specialized in GMP&GDP advisory and consultancy services for healthcare and pharmaceutical sectors. Nowadays, our company is made up by a team of 80 experienced professionals that guarantees the highest quality customer service experien...
-
Product Biocatalyst production
Is the enzyme you are interested in not commercially available and a production needs to be developed? Enzymicals has extensive expertise with scale-up from lab to pilot-scale and technology transfers to industrial manufacturing sites. We can supply your protein by in-house production up to the kg-scale...
-
Product Value-added generic development
Avivia offers full product development services to generic drug companies and specialty pharma focused on the development of new improved versions (value-added generics or generic plus) of established drug products.
Although in general the overall aim of value-added generic development projects is focu...
-
Product GMP-Support: Qualification / Validation / Consulting
Quasaar does not see itself as "processing office" of service contracts.
As an outsourcing partner, Quasaar also focuses on problem solving, increasing efficiency and process optimization, including on-site consulting and project management.
Our expertise includes i.a. following topics: ...
-
Product ANDA and NDA Technical Support
As part of its commitment to full service support, Avéma Contract Services is pleased to offer our customers support with ANDA and NDA applications, including bio equivalency studies, collecting background data, supervising technology tranfers and reviewing applications to make sure that companies com...
-
Product COMPLIANCE
AUDITING: - Support its clients on Vendor Audit; COMMISSIONING:- Manages and performs commissioning activities for new systems, equipments and facilities;
TESTING:- Performs the following testing activities using its own instrumentation saving costs and time on tests planning;
COMP...
-
Product PeP11 LabCal Excel Platform
PeP11 LabCal offers a Single Validation SaaS Solution Platform that manages all Excel spreadsheets, eliminating the need for individual validations. This comprehensive electronic Excel solution features an Electronic Signature, Audit Trail, and Role-Based Access, rendering paper signatures unnecessary ...
-
Product Audits
Due to recent regulatory developments concerning raw materials, suppliers, manufacturing and distribution, auditing has become a task of critical importance for Life Science businesses.PQE Group supports Pharma and MD companies in performing many types of certified audits, from routine monitoring to due di...
-
Product AI and non-AI validation technologies for Life Science companies
We specialize in IA and non-AI Computer System Validation, equipment validation, utilities qualification, and IT/OT infrastructure qualification. Our services comply with the regulatory standards of the EMA, FDA, WHO, and other key Life Science authorities across Europe and the Americas. With a proven trac...
-
Product Reference Materials Management Services
To support you in bringing even safer medicines to the market, we go far beyond supplying you with the right reference standards. From impurity profiling and working standards outsourcing services, all the way to managing inventory and logistics of your reference standards, we’re fully equipped to provide ...
-
Product Compliance Management
PharmSol does not limit itself only to offering Audits, but we expand our solution offerings to ensure complete Compliance at the client’s site. Towards obtaining GMP Certification from Europe, United States or WHO, PharmSol has a very effective, systematic and rational approach in providing solutions effi...
-
Product Pharmacovigilance - PvEdge - AI Enabled Drug Safety Database Suite
AI-enabled, cloud-ready,360 pharmacovigilance solution for safety database management. An end-to-end globally compliant safety software for Drugs, Devices, Vaccines and Combinational products from case intake to submissions and reporting. • Case Intake • Case Processing • Case Submission • Risk Manage...
-
Product Auditing
Our pharmaceutical auditing and management services give you a transparent view of your supply chain enabling you to identify and mitigate the intrinsic risk in your operations, supply chains and business processes. Through our shared audit programs, delivered by our global network of specialist audit...
-
Product Laboratory Services
Resource for optimal tableting: Medelpharm opened its brand new built-in laboratory in Spring 2017. The lab is under controlled atmosphere to perform powder characterisation and analysis. It houses amongst the latest innovative measuring equipment, the STYL'One Evolution Tableting Instrument, Smart test 50...
-
Product Spray Drying - GMP, Cyto or Non-Cyto, Potent
CritiTech's range of Spray Drying services is what sets it apart from other CDMOs. From Proof of Concept through GMP scale production. Dedicated Cytotoxic and Non-Cytotoxic SD equipment and facilities. Ability to process potent materials. Other key features include:
• Spray-dried dispersions - API + p...
-
Product RESEARCH AND DEVELOPMENT
We offer research and development services at all stages of the production of capsules, from idea to finished product for medicinal products, medical devices, food supplements, cosmetics and veterinary products.
We offer our clients a proper identification of their needs and requirements, r...
-
Product Digital Enablers
Achieve digital success, with specific business analysis and strategies helping your business adopting new technology as a sure competitive factor for your business today, and in the future. Our solutions: • Business Analysis • Data Lake Assessment • Data Lake Strategy & Design • Cloud Adoptio...
-
Product HVAC , DUCTING AND BMS
IN HOUSE TECHNICAL STRENGTH CONCEPTUAL-BASIC-DETAIL DESIGN ERECTION-COMMISSIONING-VALIDATION COMPLIANCE-BMS 21 CFR PART 11
AHU –DH UPTO 35% EFFECTIVE LATENT COOLING HIGH ENERGY PERFORMANCE LOW CAPITAL INVESTMENT
CHILLER COOLI...
-
Product Consulting and advisory services for gmp, risk analysis, new product dev...
Explicat Pharma GmbH offers consulting and advisory services for gmp, risk analysis, new product development strategypharmaceutical services. Contact us for more information.
-
Product Commissioned GMP Audits
Rephine’s commissioned audit service gives our customers the opportunity to work with us to design exclusive audits tailored to the specifics of their operations. Customers can commission various audits including:
-API Manufacture-Excipient Manufacture
-Investigational Medicinal Product (IMP) M...-comp267516.png)
-
Product COMMISSIONING & VALIDATION
CTP SYSTEM supports its customers in managing and directly performing all the Validation phases and Qualification activities for all kinds of equipment, systems and utilities in Pharmaceutical and related companies.
We have extensive experience in the preparation of Validation Master Plans at ...
-
Product Homogeneous Catalyst Precious Metal Recovery
The increased use of Homogeneous catalysts in the pharmaceutical sector for reactions such as hydrogenation, asymmetric hydrogenation, reductive amination, carbonylation, dehydrogenation, isomerisation - Mastermelt have developed Metal Scavenger recovery systems to capture precious metals from these unique...
-
Product Business Development
Looking to acquirenew customers? Explore our suite of business development and marketing services designed to drive growth and profitability for Life Science companies.For more information: www.outboundpharma.com/business-development-comp323974.png)
-
Product Scale up simulation and optimization
We provide scale up and optimization strategy. Scale up is based on experimental small scale data (by Designed experiments) and fed into our simulation system.We use physics and chemistry based modeling in our simulations (no black boxing).
Get the digital twin of your process!
Results are clear cont...
-
Product Arabmed CRO
ArabMed CRO is renowned for its expertise in conducting clinical trials across all phases, from Phase I to Phase IV, with a strong track record in drug research. The company also excels in medical device clinical investigations, demonstrating its commitment to advancing medical technologies. With a focus o...
-
Product QUALIFICATION OF DEPARTMENTS WITH CONTROLLED CONTAMINATION AND THERMAL MAPS
If the thermal mapping of warehouses and storage areas of the materials along the entire production and distribuition chain is complicated because of the heterogeneity of the environments and of the conditions (just think about the transporters), the qualification of production locals or of laboratori...
-
Product GMP Consultation
Onsite/offsite gap analysis of the quality management system for pharmaceutical companies against Chinese GMP, cGMP, Eudralex GMP, PIC/S GMP, and WHO GMP guidelines; Provide recommendations on gap-bridging; Carry out mock up audits and assist in correction of findings; Provide onsite technical support and ...
-
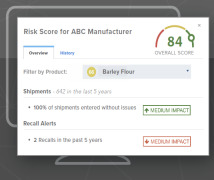
Product FDA Compliance Monitor (Quality Assurance Tool)
Automate your supply chain management. The FDA Compliance Monitor allows you to easily identify vulnerabilities in your supply chain. You can monitor the status of your own or your suppliers' FDA registration numbers as well as monitor for recalls, inspection results, warning letters, imp...
-
Product Life Science: Computer System Validation
Validation of computerized systems (CSV) according to GxP Guidelines has been a basic requirement in the life science industry for many years now. However, as the complexity of the IT landscape and the degree of automation has increased, the effort required to keep compliance has risen as well. Of cour...
-
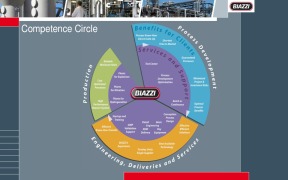
Product The Competence circle of Biazzi
BIAZZI supplies a complete know-how and engineering package as well as the related process equipment. BIAZZI offers tests in its test centre to upscale processes and make the basis for the design of the industrial plant. Biazzi is offering the delivery of the Key-Equipment/Basic Engineering or full SKID mo...
-
Product Pure steam humidifier Condair Esco
Condair Esco is a pure steam humidifier which injects plant supplied pure steam through 100% leak-proof stainless steel control valve with stainless steel steam distributors. Condensate is continuously drained from the steam distributors to prevent microbial contamination in the air stream. -
Product Inspection Readiness & Support
• Inspection Event Planning • Personnel Training and Interview Preparation • Subject Matter Expert Support • Post-Inspection Analysis
-
Product Technical Services
Complementing development sciences and clinical manufacturing, technical services include consultation on equipment specifications, scale-up, quality control, validation, and compliance auditing. On-site training is available in the fundamentals of lyophilization and validation.
&nb...
-
Product Pharmintech Exhibition
PHARMINTECH is one of the most important international exhibitions dedicated to processing, packaging and digital solutions for the Life Science industry. PHARMINTECH welcomes you in Bologna from 10th to 12th April 2019.
-
Product Pharmcovigilance
Pharmacovigilance services:
› Automated literature search and assessment
› AI supported vigilance processes
› Pharmacovigilance system
› EU-QPPV, graduated plan officer, information officer
We undertake individual pharmacovigilance tasks:
- Literature research - Evalua...
-
Product Consultancy
MFT Consult, our team combines deep expertise in regulatory compliance, facility design, QMS, and technology transfer with the ability to act quickly and efficiently. With decades of experience in the pharmaceutical and biopharmaceutical industries. we specialize in guiding startups in the pharmaceutical a...
-
Product IT/EU REGULATORY AFFAIRS - Support and management of Technical documenta...
OP Pharma has been providing innovative services in the Regulatory Affairs field for more than 20 years to small to big pharma Companies in Italy as well as in the European Union.
Our expertise covers the lifecycle management of pharmaceutical products, whole or in part, ranging from digitization ...
-
Product CONSULTING SERVICES
SciencePharma is a leading, highly experienced consultancy company providing state-of-the-art services in a field of nonclinical and clinical studies. We gather a qualified team of experts, who carry‑out tasks from the various stages of projects/studies. As the consultancy company we deal with diversity is...
-
Product Quality Compliance
For Life Science businesses in highly regulated environments, compliance is not an option. Prepare to successfully pass inspections and design post-inspections remediation plans to achieve and maintain approval for your product with PQE Group's tailored and cost-effective programs for Quality Management.
-
Product Commissioning & Qualification
Our qualified consultants strive to exceed your project goals and objectives
Our flexibility and project experiences range from the qualification of an individual piece of equipment to an entire facility.
Our PharmEng professionals have extensive experience in the development, imple...
-
Product Qualification & Engineering
The act of Validation, originally addressed only to sterilized equipment, now involves all product, process and facility matters.Choose PQE's solutions to ensure that your plant site meets your business objectives while being fully compliant with the latest Good Manufacturing Practice regulations and stand...
-
Product Regulatory & Market Access
With the growth in pharma manufacturing capability in Asia, each manufacturer nurtures an ambition to have their own footprint in the EU market.
PharmSol, with its own setups in Germany and Malta, has created a platform which provides a comprehensive, integrated and seamless market access and su...
-
Product Toxicology
Our experts ensure clients' safety by protecting them from pharmaceutical hazards
At PharmEng Technology, we understand the importance of evaluating the toxicity and safety of biopharmaceuticals, therapeutic proteins, chemicals, active pharmaceutical ingredients, and components.
P...
-
Product Regulatory Affairs
We provide solutions to meet both local and global regulatory requirements in the most cost-effective manner.
PharmEng supports Pharmaceuticals, Biologics, Over-The-Counter (OTC), Animal Health, Radiopharmaceuticals, Clinical Trial Products, Medical Devices, Combination Products, Natur...
-
Product GxP Audits and Inspection Readiness
Our auditors have real world experience supporting regulatory inspections and preparing companies for pre-approval inspections. Our team of specialists includes former regulatory agency inspectors and qualified auditors who are proficient in conducting mock inspections, internal audits, vendor and s...
-
Product Klinea Pharmaceutical Engineering
KLINEA is a company fully committed to providing added value solutions for the life sciences industry, specialized in Engineering Services, Project Management and Quality Scrutineering Tests, with offices in Madrid and Barcelona. Founded by a highly qualified technical team with more than 15 years of e...
-
Product EU/UK Qualified Person (QP) Services and MIA license
Navigating both general and country-specific regulations and requirements to supply medicinal products to the European markets can be a complex challenge for Marketing Authorization Holders (MAH). ProPharma holds both EU and UK MIA licenses which allows us to help clients overcome the complexities of su...
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance