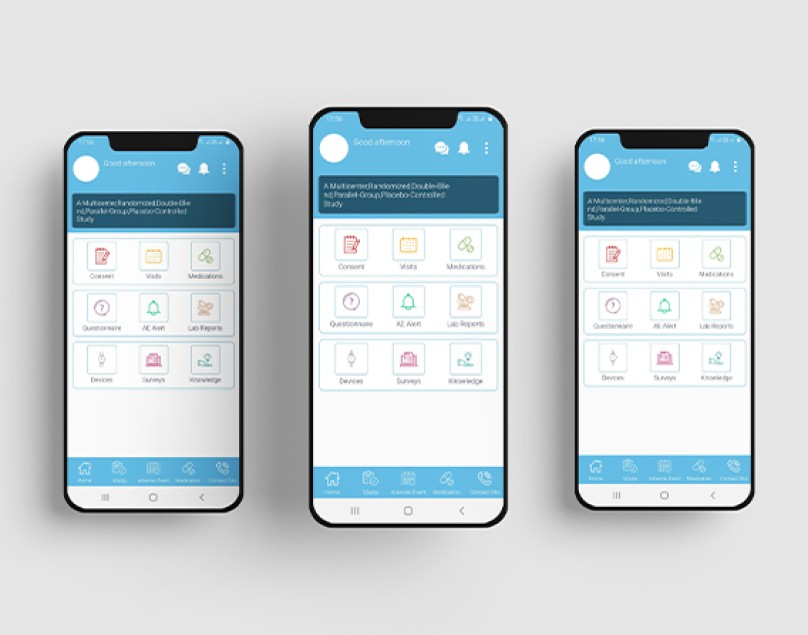
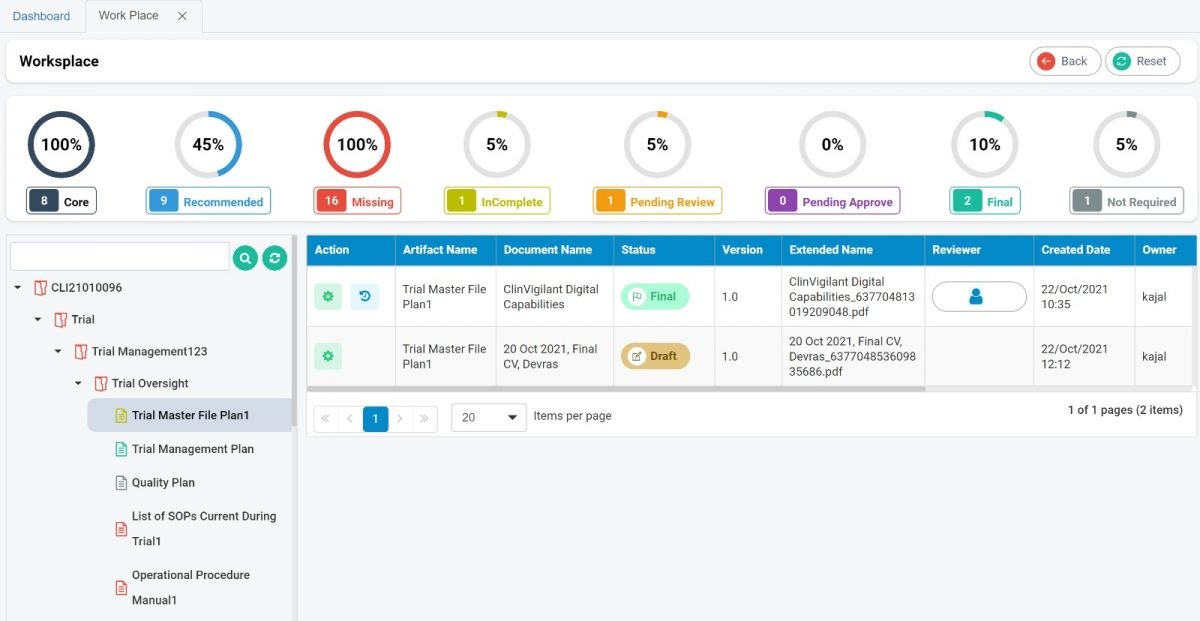
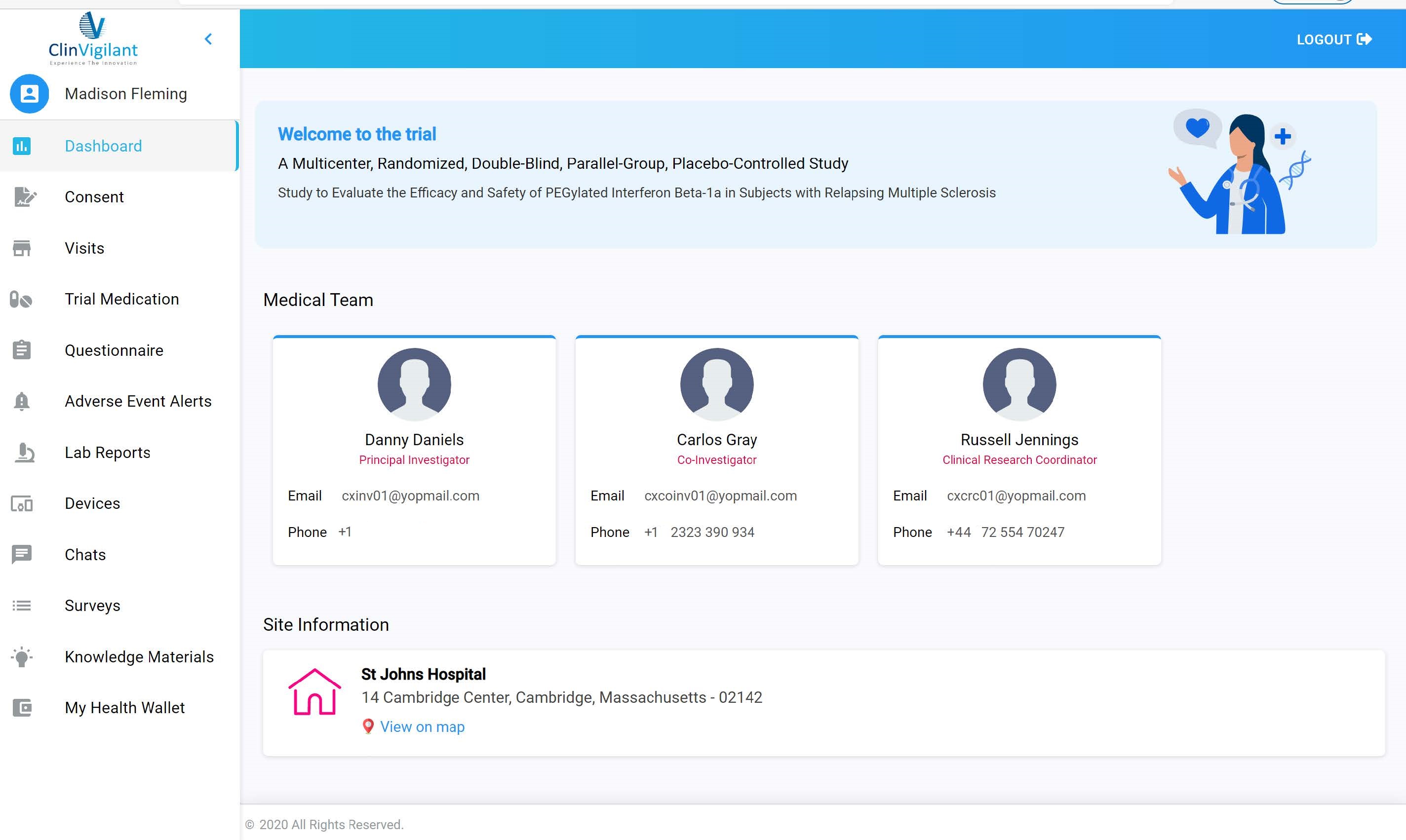
eClinical suite
Product Description
Clinvigilant Research Limited

-
GB
-
2022On CPHI since
-
4Certificates
-
25 - 49Employees
Company types
Primary activities
Categories
Specifications
Clinvigilant Research Limited

-
GB
-
2022On CPHI since
-
4Certificates
-
25 - 49Employees
Company types
Primary activities
More Products from Clinvigilant Research Limited (2)
-
Product Clinical Trial Services
Our experts will accomplish your clinical objectives by leading the way from recruitment strategies, site selection, and clinical logistics, all the way through the regulatory process.
We use our own eClinical suite to be more cost-efficient and keep you connected remotely acros... -
Product Consulting
With Clinvigilant consulting you'll get access to industry achievers that will accelerate your clinical studies timelines, reduce risks, and improve efficiency. Our strategic advisory team can help you in the following fields:
1. Regulatory Affairs > including Regula...
Clinvigilant Research Limited resources (1)
-
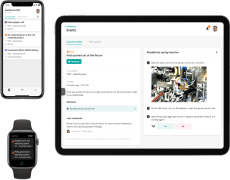
Video eClinical platform
Cloud-based eClinical suite platform for hybrid or fully decentralised trials.
Recommended Products
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance


















-comp247049.png)
























-comp318375.png)













































%20(002)-comp302826.png)

