Commercial Manufacturing Services
Product Description
Custom Pharma Services

-
GB
-
2016On CPHI since
-
250 - 499Employees
Company types
Primary activities
Categories
Specifications
Custom Pharma Services

-
GB
-
2016On CPHI since
-
250 - 499Employees
Company types
Primary activities
More Products from Custom Pharma Services (1)
-
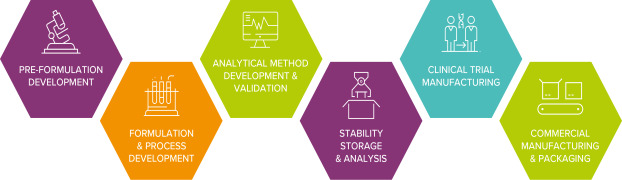
Product Clinical And Product Development Services
Pre-formulation Development
Drugs are seldom administered to patients as pure compounds, instead they are formulated into medicinal products: but many that start their development journey don’t make it to market, failing for a variety of reasons from inefficacy or toxicity to commercial or manufac...
Custom Pharma Services resources (1)
-
News Custom Pharma Services goes Back to the Future!
Whilst investing in modern technologies, Custom Pharma Services is using our decades of experience and expertise in the lifecycle management of niche and difficult to manufacture medicines to accelerate the development of new and novel medicines for our clients.
Recommended Products
Recently Visited
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance