Nasal Vaccines
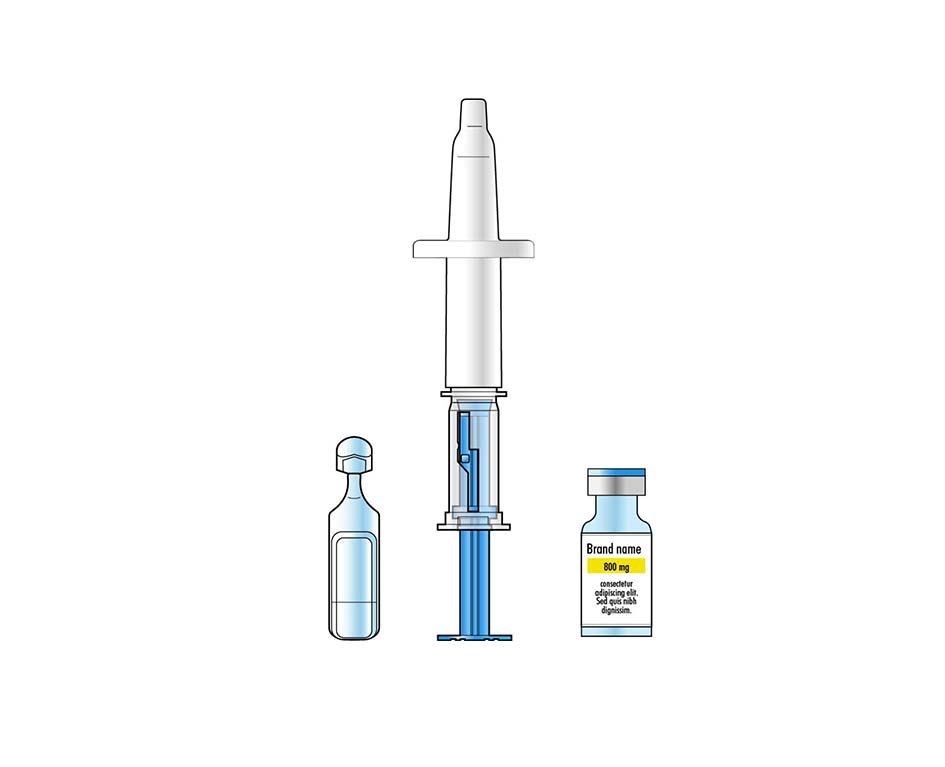
Product Description
Aptar Pharma

-
FR
-
2015On CPHI since
-
5000+Employees
Company types
Primary activities
Categories
Aptar Pharma

-
FR
-
2015On CPHI since
-
5000+Employees
Company types
Primary activities
More Products from Aptar Pharma (4)
-
Product PremiumCoat® ETFE Film Components
Aptar Pharma's PremiumCoat® offers state-of-the-art ETFE-coated stoppers and plungers, combining superior container closure integrity and compatibility. With its highest standard specifications, it ensures optimal particulate protection for sensitive drugs. -
Product UniDose (UDS) System
Aptar Pharma's Unidose systems are nasal drug delivery devices for both liquid and powder forms, enabling precise single-dose delivery without healthcare professional intervention. -
Product Bidose (BDS) System
Aptar Pharma's Bidose (BDS) is a two-shot, user-friendly nasal delivery device for liquid drugs. This sturdy, self-priming system permits accurate, one-handed operation, enabling non-invasive, systemic drug delivery without the assistance of a healthcare professional. -
Product Breath Actuated Inhaler
Aptar Pharma’s BAI is a user-friendly, breath-activated pMDI, that enhances adherence across all patient types, including children and the elderly. Suitable even for those with low inspiration rates, it ensures consistent dosage by delivering each dose upon inhalation at the mouthpiece.
Aptar Pharma resources (2)
-
Webinar Advanced Parenteral Closure Solutions, The Perfect Fit Towards Compliance With EMA GMP Annex 1 Revision
The revision of the European Medical Agency annex 1 of the GMP poses new challenges for Pharma manufacturers who have to implement a contamination control and sterility assurance strategies. Aptar Pharma Injectables’ Advanced Solutions perfectly combine to support our Pharma partners in tackling these new stringent requirements: PremiumFill®: Enhanced manufacturing means designed to deliver higher quality products with improved specifications, especially on contamination criteria, to boost your Contamination Control Strategy RTP bags: the new standard for avoiding contamination on aseptic filling lines, highly recommended in the Annex 1 revision Ready-to-Use Gamma: the only 100% guarantee of sterility at the time of use as requested in the Annex 1 revision -
Webinar Mission Possible – Sustainability in Pharmaceutical Primary Packaging
"Globally circularity is on top of consumers’ minds, and this is no different when it comes to pharmaceutical packaging. Hence, plastic materials are increasingly under scrutiny. For drug delivery systems, regulatory demand for patient safety strongly limits the use of mechanically recycled materials, and alternatives are required that make drug packaging more circular. Aptar Pharma’s Futurity™ platform of sustainable drug delivery solutions enhances circularity in pharmaceutical packaging, without compromising on performance and safety. Following the recent launch of APF Futurity™, the first metal-free, highly recyclable nasal spray pump, we are expanding our range adding the fully-recyclable Micro+ Futurity™ for dermal drug application. Created from monomaterial polyethylene, Micro+ Futurity™ leverages the user convenience and medical grade material selection of Aptar Pharma’s Airless+ dispensing solutions.
Join Aptar Pharma’s session to learn how our Futurity™ platform can strengthen your circular approach with innovative drug dispensing solutions.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







%20(1)-file145608.png)
-file143125.png)